Copper electroplating experiment
If you’re looking for copper electroplating experiment pictures information related to the copper electroplating experiment interest, you have visit the right blog. Our site always provides you with hints for viewing the highest quality video and image content, please kindly hunt and locate more enlightening video content and graphics that match your interests.
Copper Electroplating Experiment. Copper electroplating requires electricty. When current is applied to the electrolysis cell copper ii ions in solution are reduced to copper atoms at the cathode. Glass jar such as a pickle or jelly jar. An electrochemical cell is constructed to determine the efficiency of copper electroplating.
 Experiment Copper Plating An Iron Nail Free Zimsec Cambridge Revision Notes From revision.co.zw
Experiment Copper Plating An Iron Nail Free Zimsec Cambridge Revision Notes From revision.co.zw
If copper is used for the electrodes the copper anode dissolves. 3 clean nails or paper clips. How the changing of current affects the electroplating of copper. A white coating appears on the copper electrode almost immediately and after a few minutes the copper electrode has a definite zinc plating on it. Copper electroplating and faraday s law 1 purpose. Wrap one wire around the iron strip and the other around the copper strip.
How the changing of current affects the electroplating of copper.
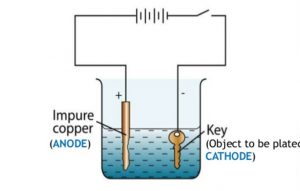
Using the scapula and weighing scale measure 5g copper sulphate crystals and add it to the water. Use one alligator clip to attach the copper electrode to the positive terminal of the battery this is now the anode and the other to attach the key to the negative terminal now called the cathode. How the changing of current affects the electroplating of copper. Using the scapula and weighing scale measure 5g copper sulphate crystals and add it to the water. This demonstration is an application of faraday s law. Copper electroplating and faraday s law 1 purpose.
 Source: examfear.com
Source: examfear.com
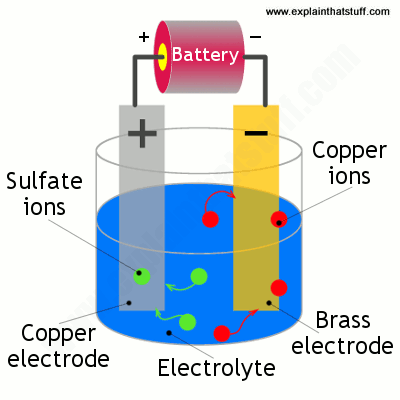
When current is applied to the electrolysis cell copper ii ions in solution are reduced to copper atoms at the cathode. Use one alligator clip to attach the copper electrode to the positive terminal of the battery this is now the anode and the other to attach the key to the negative terminal now called the cathode. If copper is used for the electrodes the copper anode dissolves. When the experiment ends the electrodes are dried and the mass of each electrode weighed on the mini balance. This demonstration is an application of faraday s law.
 Source: yenka.com
Source: yenka.com
An electrochemical cell is constructed to determine the efficiency of copper electroplating. Copper electroplating and faraday s law 1 purpose. The reaction is the reverse of the cathode reaction. Cut two 12 cm sections of wire. Table 17 8 sample data and results of calculations of a copper zinc electrolysis cell.
 Source: chemdemos.uoregon.edu
Source: chemdemos.uoregon.edu
Welcome to science at home in this experiment we are exploring copper electroplating by coating a nail with copper. Aim to find the amount copper gains or loses on the electrodes using different amounts of current each time during electrolysis. Stir copper sulfate into some hot water in a beaker until no more will dissolve. Copper electroplating requires electricty. Read the section below to perform this experiment.
 Source: researchgate.net
Source: researchgate.net
Your solution should be dark blue. Aim to find the amount copper gains or loses on the electrodes using different amounts of current each time during electrolysis. Copper atoms on the anode are oxidized to copper ii ions. Glass jar such as a pickle or jelly jar. When the experiment ends the electrodes are dried and the mass of each electrode weighed on the mini balance.
 Source: cikguwong.blogspot.com
Source: cikguwong.blogspot.com
Introduction electroplating is generally carried out in order to improve the appearance. Your solution should be dark blue. Use one alligator clip to attach the copper electrode to the positive terminal of the battery this is now the anode and the other to attach the key to the negative terminal now called the cathode. Table 17 8 sample data and results of calculations of a copper zinc electrolysis cell. Introduction copper roofing is a prominent part of campus architecture.
 Source: m.youtube.com
Source: m.youtube.com
An electrochemical cell is constructed to determine the efficiency of copper electroplating. Copper electroplating requires electricty. Introduction copper roofing is a prominent part of campus architecture. Using the scapula and weighing scale measure 5g copper sulphate crystals and add it to the water. When current is applied to the electrolysis cell copper ii ions in solution are reduced to copper atoms at the cathode.
 Source: sites.google.com
Source: sites.google.com
2 cups of vinegar. When the experiment ends the electrodes are dried and the mass of each electrode weighed on the mini balance. Copper strips 10 distilled water 2l copper sulphate 50g electronic scale 1 ziploc bag 10 procedure. Aim to find the amount copper gains or loses on the electrodes using different amounts of current each time during electrolysis. Stir copper sulfate into some hot water in a beaker until no more will dissolve.
 Source: classnotes.org.in
Source: classnotes.org.in
Wrap one wire around the iron strip and the other around the copper strip. An electrochemical cell is constructed to determine the efficiency of copper electroplating. The cathode gains mass the anode looses mass. Introduction electroplating is generally carried out in order to improve the appearance. If copper is used for the electrodes the copper anode dissolves.
 Source: docbrown.info
Source: docbrown.info
Fill the jar with vinegar. An experiment to show how electroplating using copper electrodes. Cut two 12 cm sections of wire. Introduction electroplating is generally carried out in order to improve the appearance. A white coating appears on the copper electrode almost immediately and after a few minutes the copper electrode has a definite zinc plating on it.
 Source: revision.co.zw
Source: revision.co.zw
Copper electroplating requires electricty. Welcome to science at home in this experiment we are exploring copper electroplating by coating a nail with copper. Table 17 8 sample data and results of calculations of a copper zinc electrolysis cell. Aim to find the amount copper gains or loses on the electrodes using different amounts of current each time during electrolysis. This demonstration is an application of faraday s law.
 Source: docbrown.info
Source: docbrown.info
How the changing of current affects the electroplating of copper. 1 teaspoon of salt. Cut two 12 cm sections of wire. Read the section below to perform this experiment. The cathode gains mass the anode looses mass.
 Source: gigalinsights.com
Source: gigalinsights.com
The results of this experiment can lead to a discussion about electroplating and the electrolytic refining of copper. Copper electroplating requires electricty. This demonstration is an application of faraday s law. Introduction copper roofing is a prominent part of campus architecture. 1 teaspoon of salt.
 Source: chemedx.org
Source: chemedx.org
1 teaspoon of salt. 2 cups of vinegar. Your solution should be dark blue. When the experiment ends the electrodes are dried and the mass of each electrode weighed on the mini balance. 1 teaspoon of salt.
 Source: explainthatstuff.com
Source: explainthatstuff.com
Introduction copper roofing is a prominent part of campus architecture. When the experiment ends the electrodes are dried and the mass of each electrode weighed on the mini balance. Cut two 12 cm sections of wire. Introduction electroplating is generally carried out in order to improve the appearance. Your solution should be dark blue.
 Source: chemedx.org
Source: chemedx.org
The cathode gains mass the anode looses mass. A white coating appears on the copper electrode almost immediately and after a few minutes the copper electrode has a definite zinc plating on it. Creating the electroplating solution. Chemical treatments are tested to produce a light green patina that is characteristic of aged copper. An electrochemical cell is constructed to determine the efficiency of copper electroplating.
If you find this site good, please support us by sharing this posts to your favorite social media accounts like Facebook, Instagram and so on or you can also save this blog page with the title copper electroplating experiment by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.





