Corrosion of iron experiment
If you’re searching for corrosion of iron experiment pictures information linked to the corrosion of iron experiment interest, you have visit the right site. Our website frequently provides you with hints for viewing the maximum quality video and image content, please kindly surf and locate more enlightening video articles and images that match your interests.
Corrosion Of Iron Experiment. Recall that phenolphthalein turns pink in the presence of hydroxide and ferricyanide turns a deep blue in the presence of iron ii rust. Background the importance of corrosion can be seen in daily life. Rates of corrosion can be affected by many factors including the environment stress oxygen availability etc. Corrosion of iron experiment purpose observe the corrosion of iron and investigate conditions related to corrosion.
 Rusting Of Iron Consequence Chemistry Prevention Example And Videos From toppr.com
Rusting Of Iron Consequence Chemistry Prevention Example And Videos From toppr.com
In nature the oxidizing agent is frequently atmospheric oxygen dissolved in water. Place an iron nail in one of the test tubes. Corrosion of iron experiment. Background the importance of corrosion can be seen in daily life. Corrosion causes accidents in industry on highways and in homes. Objective the objective of this experiment is to measure the corrosion rate of two different metals and to show the effectiveness of the use of inhibitors to protect metals from corrosion.
This is a long term corrosion experiment requiring a few minutes each day first for setup then for observation.
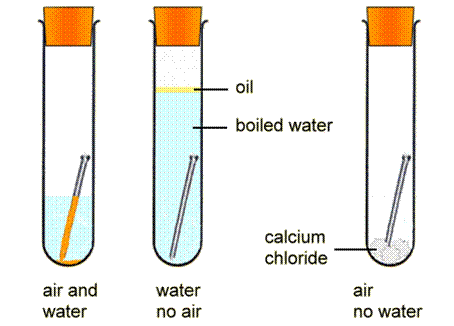
Recall that phenolphthalein turns pink in the presence of hydroxide and ferricyanide turns a deep blue in the presence of iron ii rust. Corrosion is an oxidation reduction redox reaction in which a free metal is oxidized or corroded by some oxidizing agent. Use this class experiment to help students investigate what conditions are needed for the familiar process of rusting in this practical students put iron nails in various conditions including wet dry air free and salty to find out what causes iron to rust. The collapse of the silver bridge in 1967 and the mianus river bridge in 1983 is attributed to the corrosion of the steel iron components of the bridge. Clean the nails with sand paper. An understanding of the activity series investigated in experiment 6 suggests that one way of preventing the corrosion of iron is to protect it with a more active metal.
 Source: chembam.com
Source: chembam.com
The collapse of the silver bridge in 1967 and the mianus river bridge in 1983 is attributed to the corrosion of the steel iron components of the bridge. Objective the objective of this experiment is to measure the corrosion rate of two different metals and to show the effectiveness of the use of inhibitors to protect metals from corrosion. Rates of corrosion can be affected by many factors including the environment stress oxygen availability etc. The collapse of the silver bridge in 1967 and the mianus river bridge in 1983 is attributed to the corrosion of the steel iron components of the bridge. In nature the oxidizing agent is frequently atmospheric oxygen dissolved in water.
 Source: docbrown.info
Source: docbrown.info
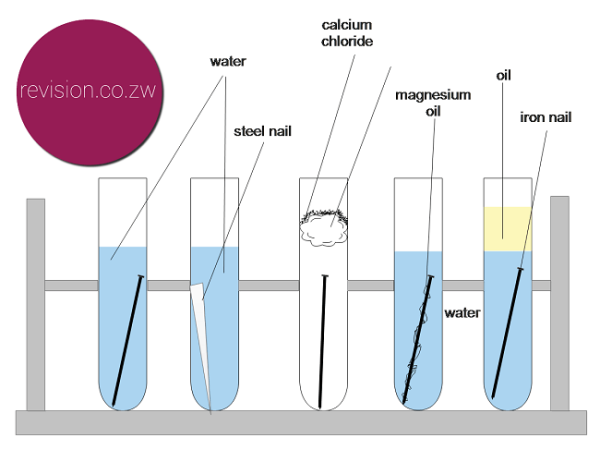
Ferroxyl indicator turns blue when iron rusts iron nails sand paper magnesium ribbon zinc copper five test tubes paint method. Rates of corrosion can be affected by many factors including the environment stress oxygen availability etc. An understanding of the activity series investigated in experiment 6 suggests that one way of preventing the corrosion of iron is to protect it with a more active metal. Many buildings made up of reinforced concrete also undergo structural failures over long periods of time due to rusting. Ferroxyl indicator turns blue when iron rusts iron nails sand paper magnesium ribbon zinc copper five test tubes paint method.
 Source: bajraionline.com
Source: bajraionline.com
Students will be able to observe the results of corrosion over several days up to 2 weeks. Place ferroxyl indicator to a depth of about 5cm in five test tubes. Many buildings made up of reinforced concrete also undergo structural failures over long periods of time due to rusting. In this experiment we can watch the corrosion reaction by using substances that produce a color change when they react with the products of the iron oxidation or oxygen reduction. The collapse of the silver bridge in 1967 and the mianus river bridge in 1983 is attributed to the corrosion of the steel iron components of the bridge.
 Source: berryberryeasy.com
Source: berryberryeasy.com
Another way to prevent the corrosion of iron is to exclude oxygen and moisture from its surface with a protective coating. Rates of corrosion can be affected by many factors including the environment stress oxygen availability etc. An understanding of the activity series investigated in experiment 6 suggests that one way of preventing the corrosion of iron is to protect it with a more active metal. To investigate the corrosion of iron materials. In nature the oxidizing agent is frequently atmospheric oxygen dissolved in water.
 Source: brainly.in
Source: brainly.in
Pre lab discussion corrosion is a oxidation reduction redox reaction in which a free metal is oxidized or corroded by some oxidizing agent. To investigate the corrosion of iron materials. Ferroxyl indicator turns blue when iron rusts iron nails sand paper magnesium ribbon zinc copper five test tubes paint method. Recall that phenolphthalein turns pink in the presence of hydroxide and ferricyanide turns a deep blue in the presence of iron ii rust. In nature the oxidizing agent is frequently atmospheric oxygen dissolved in water.
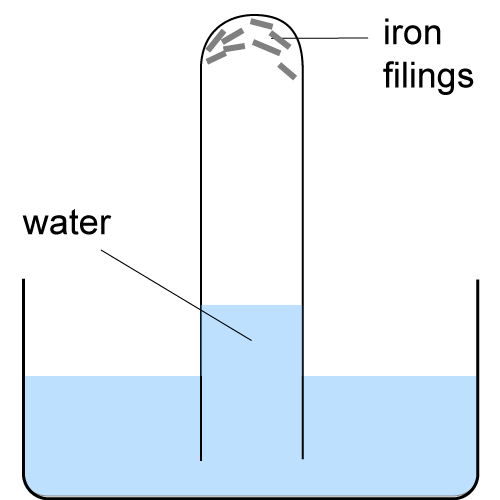 Source: gradegorilla.com
Source: gradegorilla.com
Clean the nails with sand paper. In nature the oxidizing agent is frequently atmospheric oxygen dissolved in water. Students will be able to observe the results of corrosion over several days up to 2 weeks. Rates of corrosion can be affected by many factors including the environment stress oxygen availability etc. To investigate the corrosion of iron materials.
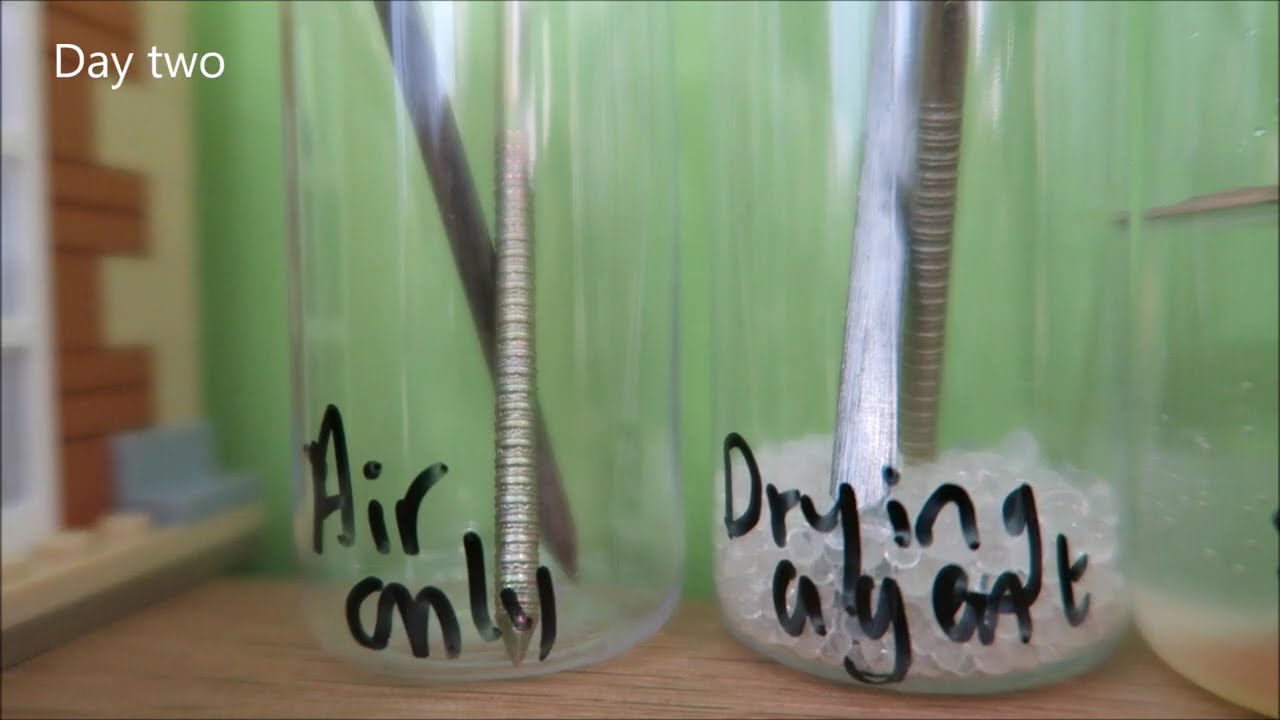 Source: m.youtube.com
Source: m.youtube.com
Ap chemistry oxidation reduction corrosion of iron experiment purpose observe the corrosion of iron and investigate conditions related to corrosion. The collapse of the silver bridge in 1967 and the mianus river bridge in 1983 is attributed to the corrosion of the steel iron components of the bridge. Objective the objective of this experiment is to measure the corrosion rate of two different metals and to show the effectiveness of the use of inhibitors to protect metals from corrosion. Another way to prevent the corrosion of iron is to exclude oxygen and moisture from its surface with a protective coating. Rusted iron can be a breeding ground for bacteria that cause tetanus.
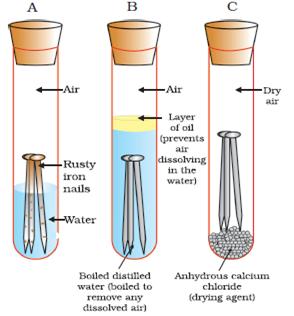 Source: topperlearning.com
Source: topperlearning.com
An understanding of the activity series investigated in experiment 6 suggests that one way of preventing the corrosion of iron is to protect it with a more active metal. Pre lab discussion corrosion is a oxidation reduction redox reaction in which a free metal is oxidized or corroded by some oxidizing agent. Use this class experiment to help students investigate what conditions are needed for the familiar process of rusting in this practical students put iron nails in various conditions including wet dry air free and salty to find out what causes iron to rust. In this experiment we can watch the corrosion reaction by using substances that produce a color change when they react with the products of the iron oxidation or oxygen reduction. Rates of corrosion can be affected by many factors including the environment stress oxygen availability etc.
 Source: meritnation.com
Source: meritnation.com
Pre lab discussion corrosion is a oxidation reduction redox reaction in which a free metal is oxidized or corroded by some oxidizing agent. Ap chemistry oxidation reduction corrosion of iron experiment purpose observe the corrosion of iron and investigate conditions related to corrosion. The collapse of the silver bridge in 1967 and the mianus river bridge in 1983 is attributed to the corrosion of the steel iron components of the bridge. Place ferroxyl indicator to a depth of about 5cm in five test tubes. Place an iron nail in one of the test tubes.
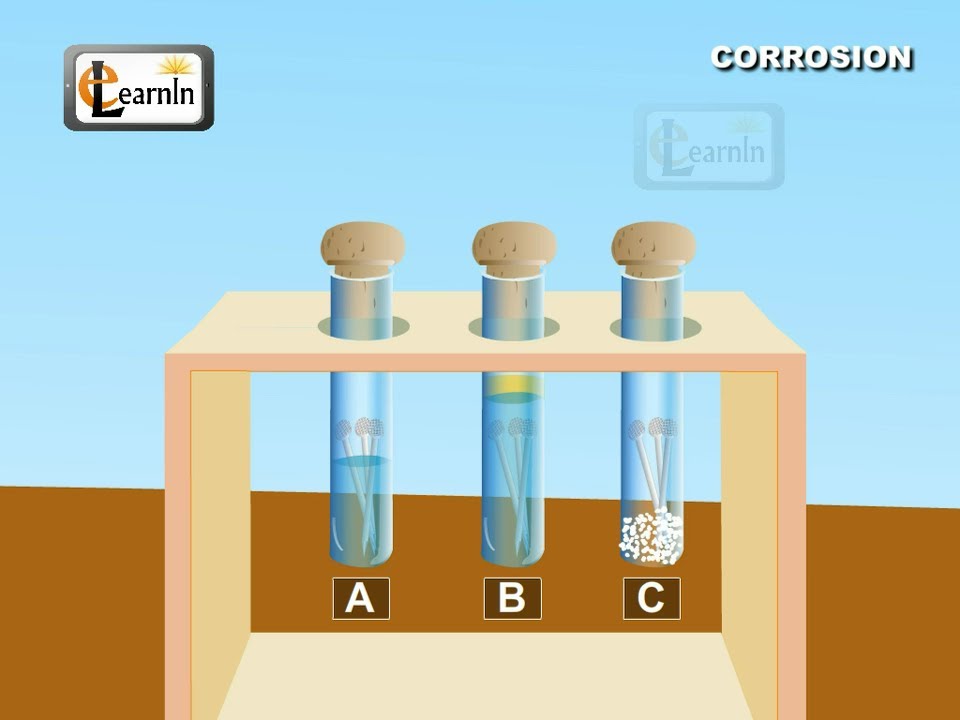 Source: youtube.com
Source: youtube.com
Background the importance of corrosion can be seen in daily life. Rates of corrosion can be affected by many factors including the environment stress oxygen availability etc. Corrosion causes accidents in industry on highways and in homes. In nature the oxidizing agent is frequently atmospheric oxygen dissolved in water. Recall that phenolphthalein turns pink in the presence of hydroxide and ferricyanide turns a deep blue in the presence of iron ii rust.
 Source: revision.co.zw
Source: revision.co.zw
Ap chemistry oxidation reduction corrosion of iron experiment purpose observe the corrosion of iron and investigate conditions related to corrosion. Corrosion is an oxidation reduction redox reaction in which a free metal is oxidized or corroded by some oxidizing agent. Corrosion causes accidents in industry on highways and in homes. Corrosion of iron experiment purpose observe the corrosion of iron and investigate conditions related to corrosion. Place ferroxyl indicator to a depth of about 5cm in five test tubes.

Ferroxyl indicator turns blue when iron rusts iron nails sand paper magnesium ribbon zinc copper five test tubes paint method. In nature the oxidizing agent is frequently atmospheric oxygen dissolved in water. Corrosion is an oxidation reduction redox reaction in which a free metal is oxidized or corroded by some oxidizing agent. Students will be able to observe the results of corrosion over several days up to 2 weeks. Pre lab discussion corrosion is a oxidation reduction redox reaction in which a free metal is oxidized or corroded by some oxidizing agent.
 Source: toppr.com
Source: toppr.com
Recall that phenolphthalein turns pink in the presence of hydroxide and ferricyanide turns a deep blue in the presence of iron ii rust. In nature the oxidizing agent is frequently atmospheric oxygen dissolved in water. Ap chemistry oxidation reduction corrosion of iron experiment purpose observe the corrosion of iron and investigate conditions related to corrosion. Observe the corrosion of iron and investigate conditions related to corrosion. Rates of corrosion can be affected by many factors including the environment stress oxygen availability etc.
 Source: olc.edu.hk
Source: olc.edu.hk
Pre lab discussion corrosion is a oxidation reduction redox reaction in which a free metal is oxidized or corroded by some oxidizing agent. The collapse of the silver bridge in 1967 and the mianus river bridge in 1983 is attributed to the corrosion of the steel iron components of the bridge. Ap chemistry oxidation reduction corrosion of iron experiment purpose observe the corrosion of iron and investigate conditions related to corrosion. Rusted iron can be a breeding ground for bacteria that cause tetanus. Use this class experiment to help students investigate what conditions are needed for the familiar process of rusting in this practical students put iron nails in various conditions including wet dry air free and salty to find out what causes iron to rust.
 Source: ppsks3.blogspot.com
Source: ppsks3.blogspot.com
Recall that phenolphthalein turns pink in the presence of hydroxide and ferricyanide turns a deep blue in the presence of iron ii rust. Recall that phenolphthalein turns pink in the presence of hydroxide and ferricyanide turns a deep blue in the presence of iron ii rust. Ferroxyl indicator turns blue when iron rusts iron nails sand paper magnesium ribbon zinc copper five test tubes paint method. Clean the nails with sand paper. Place ferroxyl indicator to a depth of about 5cm in five test tubes.
If you find this site value, please support us by sharing this posts to your favorite social media accounts like Facebook, Instagram and so on or you can also save this blog page with the title corrosion of iron experiment by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.





