Electrolysis of salt water experiment
If you’re searching for electrolysis of salt water experiment pictures information related to the electrolysis of salt water experiment keyword, you have come to the right site. Our website frequently gives you hints for viewing the highest quality video and image content, please kindly surf and locate more informative video content and images that fit your interests.
Electrolysis Of Salt Water Experiment. In water salt actually splits into na and cl ions which are very good at carrying current or the flow of electric charges. Reduction of na e 2 7 v is energetically more difficult than the reduction of water 1 23 v so in aqueous solution the latter will prevail. Lemon juice is a decent electrolyte because it is an acid. So the electrolysis of wcl4 produces w and cl2.
 Pin On Stem From id.pinterest.com
Pin On Stem From id.pinterest.com
Hydrogen gas will be seen to bubble up at the cathode and chlorine gas will bubble at the anode. In water salt actually splits into na and cl ions which are very good at carrying current or the flow of electric charges. Lemon juice is a decent electrolyte because it is an acid. In this experiment you will be taking a sample of salt water and adding a flow of. Reduction of na e 2 7 v is energetically more difficult than the reduction of water 1 23 v so in aqueous solution the latter will prevail. In water salt actually splits into na and cl ions which are very good at carrying current or the flow of electric charges.
Lemon juice is a decent electrolyte because it is an acid.
Electrolysis is a technique used by scientists to separate a compound or molecule into its component parts. Hydrogen gas will be seen to bubble up at the cathode and chlorine gas will bubble at the anode. Electrolysis is a technique used by scientists to separate a compound or molecule into its component parts. By adding electricity to water and providing a path for the different particles to follow water can be separated into hydrogen and oxygen. So the electrolysis of wcl4 produces w and cl2. Nacl aq can be reliably electrolysed to produce hydrogen.

Electrolysis of an aqueous solution of table salt nacl or sodium chloride produces aqueous sodium hydroxide and chlorine although usually only in minute amounts. Electrolysis of an aqueous solution of table salt nacl or sodium choride produces aqueous sodium hydroxide and chlorine although usually only in minute amounts. So the electrolysis of wcl4 produces w and cl2. In this experiment you will be taking a sample of salt water and adding a flow of. Hydrogen gas will be seen to bubble up at the cathode and chlorine gas will bubble at the anode.
 Source: scienceprojectideas.org
Source: scienceprojectideas.org
Electrolysis of an aqueous solution of table salt nacl or sodium chloride produces aqueous sodium hydroxide and chlorine although usually only in minute amounts. By adding electricity to water and providing a path for the different particles to follow water can be separated into hydrogen and oxygen. So the electrolysis of wcl4 produces w and cl2. Deduce the products of the electrolysis of a molten salt electrolysis of a molten salt produces the elements from the salt. Nacl aq can be reliably electrolysed to produce hydrogen.

In the water salt solution the gases that are produced are hydrogen oxygen and chlorine gas cl 2 you don t want to inhale this stuff. In the water salt solution the gases that are produced are hydrogen oxygen and chlorine gas cl 2 you don t want to inhale this stuff. Deduce the products of the electrolysis of a molten salt electrolysis of a molten salt produces the elements from the salt. So the electrolysis of wcl4 produces w and cl2. Electrolysis of an aqueous solution of table salt nacl or sodium chloride produces aqueous sodium hydroxide and chlorine although usually only in minute amounts.
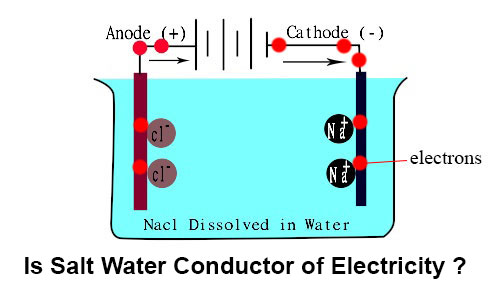 Source: ekunji.com
Source: ekunji.com
So the electrolysis of wcl4 produces w and cl2. Deduce the products of the electrolysis of a molten salt electrolysis of a molten salt produces the elements from the salt. Electrolysis of an aqueous solution of table salt nacl or sodium choride produces aqueous sodium hydroxide and chlorine although usually only in minute amounts. So the electrolysis of wcl4 produces w and cl2. By adding electricity to water and providing a path for the different particles to follow water can be separated into hydrogen and oxygen.
 Source: studylib.net
Source: studylib.net
Reduction of na e 2 7 v is energetically more difficult than the reduction of water 1 23 v so in aqueous solution the latter will prevail. Electrolysis of an aqueous solution of table salt nacl or sodium choride produces aqueous sodium hydroxide and chlorine although usually only in minute amounts. Lemon juice is a decent electrolyte because it is an acid. Reduction of na e 2 7 v is energetically more difficult than the reduction of water 1 23 v so in aqueous solution the latter will prevail. Nacl aq can be reliably electrolysed to produce hydrogen.
 Source: youtube.com
Source: youtube.com
In water salt actually splits into na and cl ions which are very good at carrying current or the flow of electric charges. Deduce the products of the electrolysis of a molten salt electrolysis of a molten salt produces the elements from the salt. Hydrogen gas will be seen to bubble up at the cathode and chlorine gas will bubble at the anode. Electrolysis of water salt easy science fair project for school like it and comments below and don t forget t. In the water salt solution the gases that are produced are hydrogen oxygen and chlorine gas cl 2 you don t want to inhale this stuff.
 Source: id.pinterest.com
Source: id.pinterest.com
Electrolysis of water salt easy science fair project for school like it and comments below and don t forget t. By adding electricity to water and providing a path for the different particles to follow water can be separated into hydrogen and oxygen. In this experiment you will be taking a sample of salt water and adding a flow of. Lemon juice is a decent electrolyte because it is an acid. Electrolysis of water.
 Source: revisionscience.com
Source: revisionscience.com
Lemon juice is a decent electrolyte because it is an acid. Hi guys watch this amazing video experiment. Electrolysis of water. Hydrogen gas will be seen to bubble up at the cathode and chlorine gas will bubble at the anode. Hydrogen gas will be seen to bubble up at the cathode and chlorine gas will bubble at the anode.
 Source: orbitingfrog.com
Source: orbitingfrog.com
Deduce the products of the electrolysis of a molten salt electrolysis of a molten salt produces the elements from the salt. In water salt actually splits into na and cl ions which are very good at carrying current or the flow of electric charges. Electrolysis of water. Nacl aq can be reliably electrolysed to produce hydrogen. Electrolysis of an aqueous solution of table salt nacl or sodium choride produces aqueous sodium hydroxide and chlorine although usually only in minute amounts.

Electrolysis of water. Lemon juice is a decent electrolyte because it is an acid. Reduction of na e 2 7 v is energetically more difficult than the reduction of water 1 23 v so in aqueous solution the latter will prevail. So the electrolysis of wcl4 produces w and cl2. In the water salt solution the gases that are produced are hydrogen oxygen and chlorine gas cl 2 you don t want to inhale this stuff.
 Source: docbrown.info
Source: docbrown.info
Nacl aq can be reliably electrolysed to produce hydrogen. Electrolysis of water salt easy science fair project for school like it and comments below and don t forget t. Hydrogen gas will be seen to bubble up at the cathode and chlorine gas will bubble at the anode. Nacl aq can be reliably electrolysed to produce hydrogen. Lemon juice is a decent electrolyte because it is an acid.
 Source: youtube.com
Source: youtube.com
In water salt actually splits into na and cl ions which are very good at carrying current or the flow of electric charges. Nacl aq can be reliably electrolysed to produce hydrogen. In this experiment you will be taking a sample of salt water and adding a flow of. Reduction of na e 2 7 v is energetically more difficult than the reduction of water 1 23 v so in aqueous solution the latter will prevail. In the water salt solution the gases that are produced are hydrogen oxygen and chlorine gas cl 2 you don t want to inhale this stuff.
 Source: thejoysharing.com
Source: thejoysharing.com
By adding electricity to water and providing a path for the different particles to follow water can be separated into hydrogen and oxygen. In water salt actually splits into na and cl ions which are very good at carrying current or the flow of electric charges. In this experiment you will be taking a sample of salt water and adding a flow of. In water salt actually splits into na and cl ions which are very good at carrying current or the flow of electric charges. Hydrogen gas will be seen to bubble up at the cathode and chlorine gas will bubble at the anode.
 Source: docbrown.info
Source: docbrown.info
Nacl aq can be reliably electrolysed to produce hydrogen. So the electrolysis of wcl4 produces w and cl2. In the water salt solution the gases that are produced are hydrogen oxygen and chlorine gas cl 2 you don t want to inhale this stuff. Deduce the products of the electrolysis of a molten salt electrolysis of a molten salt produces the elements from the salt. Reduction of na e 2 7 v is energetically more difficult than the reduction of water 1 23 v so in aqueous solution the latter will prevail.
 Source: youtube.com
Source: youtube.com
Deduce the products of the electrolysis of a molten salt electrolysis of a molten salt produces the elements from the salt. In this experiment you will be taking a sample of salt water and adding a flow of. Electrolysis is a technique used by scientists to separate a compound or molecule into its component parts. Hydrogen gas will be seen to bubble up at the cathode and chlorine gas will bubble at the anode. Electrolysis of an aqueous solution of table salt nacl or sodium choride produces aqueous sodium hydroxide and chlorine although usually only in minute amounts.
If you find this site helpful, please support us by sharing this posts to your preference social media accounts like Facebook, Instagram and so on or you can also save this blog page with the title electrolysis of salt water experiment by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.





