Electrolysis of water explained
If you’re searching for electrolysis of water explained pictures information connected with to the electrolysis of water explained interest, you have pay a visit to the ideal blog. Our website frequently provides you with suggestions for seeking the highest quality video and image content, please kindly hunt and locate more enlightening video content and graphics that fit your interests.
Electrolysis Of Water Explained. Electrolysis of water. Electrolysis of water is the process of using electricity to decompose water into oxygen and hydrogen gas. Electrolysis is the chemical process of using an electrical current to stimulate non spontaneous reactions. Battery 1 per group.
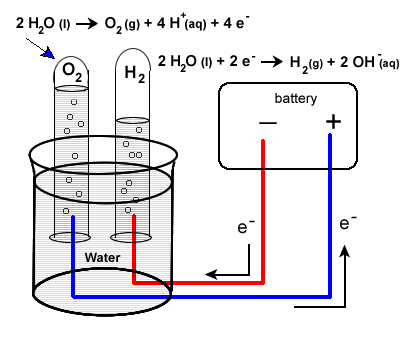 16 7 Electrolysis Using Electricity To Do Chemistry Chemistry Libretexts From chem.libretexts.org
16 7 Electrolysis Using Electricity To Do Chemistry Chemistry Libretexts From chem.libretexts.org
Electrolysis of water. Electrolysis of water experiment energy is stored in the bonds of molecules. Compound can be extracted from water electrolysis will be able to explain how energy hydrogen flows through the electrolysis system molecule oxygen. Battery 1 per group. What is electrolysis reactions chemistry fuseschoolelectrolysis is electrical current flow through a liquid which causes chemical changes. A non spontaneous reaction is one that needs energy to work while it proceeds.
Battery 1 per group.
A non spontaneous reaction is one that needs energy to work while it proceeds. Battery 1 per group. When these bonds split apart the energy released can be used to do work. Hydrogen gas released in this way can be used as hydrogen fuel or remixed with the oxygen to create oxyhydrogen gas which is used in welding and other applications. What is electrolysis reactions chemistry fuseschoolelectrolysis is electrical current flow through a liquid which causes chemical changes. Will be able to explain how hydrogen.
 Source: brainly.in
Source: brainly.in
Will be able to explain how hydrogen. What is electrolysis reactions chemistry fuseschoolelectrolysis is electrical current flow through a liquid which causes chemical changes. Electrolysis of water experiment energy is stored in the bonds of molecules. Compound can be extracted from water electrolysis will be able to explain how energy hydrogen flows through the electrolysis system molecule oxygen. Electric current being passed through the water.
 Source: pinterest.com
Source: pinterest.com
Compound can be extracted from water electrolysis will be able to explain how energy hydrogen flows through the electrolysis system molecule oxygen. Electrolysis of water. Electrolysis is the chemical process of using an electrical current to stimulate non spontaneous reactions. Electrolysis of water experiment energy is stored in the bonds of molecules. Electric current being passed through the water.
 Source: researchgate.net
Source: researchgate.net
Will be able to explain how hydrogen. A non spontaneous reaction is one that needs energy to work while it proceeds. Electrolysis of water is the decomposition of water h2o into oxygen o2 and hydrogen gas h2 due to an. Electrolysis of water. When these bonds split apart the energy released can be used to do work.
 Source: chem.libretexts.org
Source: chem.libretexts.org
Battery 1 per group. What is electrolysis reactions chemistry fuseschoolelectrolysis is electrical current flow through a liquid which causes chemical changes. Electrolysis of water produces hydrogen gas at the cathode which contains a lower proportion of deuterium than the original water. Applications since hydrogen can be produced more affordably from fossil fuels. Electrolysis of water is the process of using electricity to decompose water into oxygen and hydrogen gas.
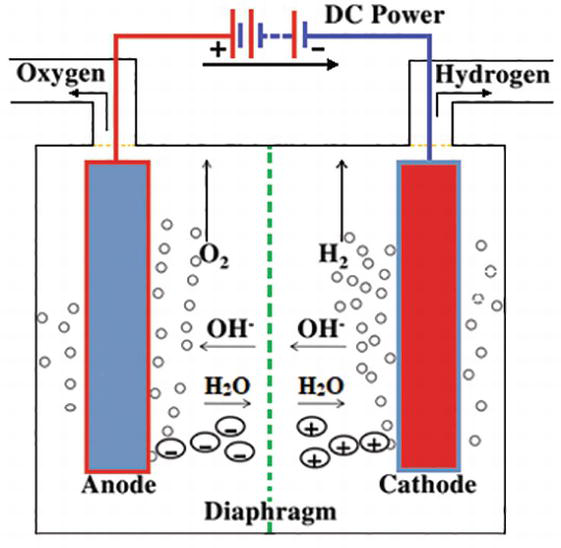 Source: intechopen.com
Source: intechopen.com
The isotope effect stems from the differences in the rates of dissociation of a proton h and a deuteron d from water and the rates of neutralization of these hydrated ions and thus has a kinetic basis. Electrolysis of water produces hydrogen gas at the cathode which contains a lower proportion of deuterium than the original water. Will be able to explain how hydrogen. What is electrolysisthe electrolytic process of deposition of superior metal on the surface of the base metal or article is known as electro platingit makes. When these bonds split apart the energy released can be used to do work.
 Source: researchgate.net
Source: researchgate.net
Electrolysis of water. Electrolysis of water is the process of using electricity to decompose water into oxygen and hydrogen gas. Electrolysis is the chemical process of using an electrical current to stimulate non spontaneous reactions. Will be able to explain how hydrogen. Breaking apart liquid water molecules into hydrogen and oxygen gas creates an enormous amount of energy which can be turned into useful electricity to power our homes and cars.
 Source: youtube.com
Source: youtube.com
Electrolysis of water produces hydrogen gas at the cathode which contains a lower proportion of deuterium than the original water. Breaking apart liquid water molecules into hydrogen and oxygen gas creates an enormous amount of energy which can be turned into useful electricity to power our homes and cars. Electric current being passed through the water. When these bonds split apart the energy released can be used to do work. Electrolysis of water is the decomposition of water h2o into oxygen o2 and hydrogen gas h2 due to an.

Electrolysis of water. Electrolysis of water is the decomposition of water h2o into oxygen o2 and hydrogen gas h2 due to an. Electric current being passed through the water. Electrolysis of water experiment energy is stored in the bonds of molecules. Electrolysis of water produces hydrogen gas at the cathode which contains a lower proportion of deuterium than the original water.
 Source: sciencedirect.com
Source: sciencedirect.com
A non spontaneous reaction is one that needs energy to work while it proceeds. Electrolysis of water. Hydrogen gas released in this way can be used as hydrogen fuel or remixed with the oxygen to create oxyhydrogen gas which is used in welding and other applications. Breaking apart liquid water molecules into hydrogen and oxygen gas creates an enormous amount of energy which can be turned into useful electricity to power our homes and cars. The isotope effect stems from the differences in the rates of dissociation of a proton h and a deuteron d from water and the rates of neutralization of these hydrated ions and thus has a kinetic basis.
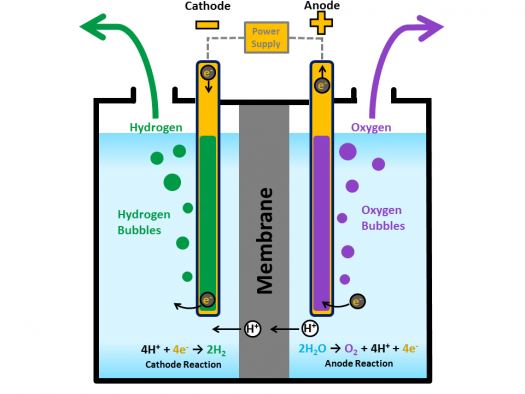 Source: energy.gov
Source: energy.gov
This electrolytic process is rarely used in industrial. The isotope effect stems from the differences in the rates of dissociation of a proton h and a deuteron d from water and the rates of neutralization of these hydrated ions and thus has a kinetic basis. Sometimes called water splitting electrolysis requires a minimum potential difference of 1 23 volts. Electrolysis of water is the decomposition of water h2o into oxygen o2 and hydrogen gas h2 due to an. Electrolysis is the chemical process of using an electrical current to stimulate non spontaneous reactions.
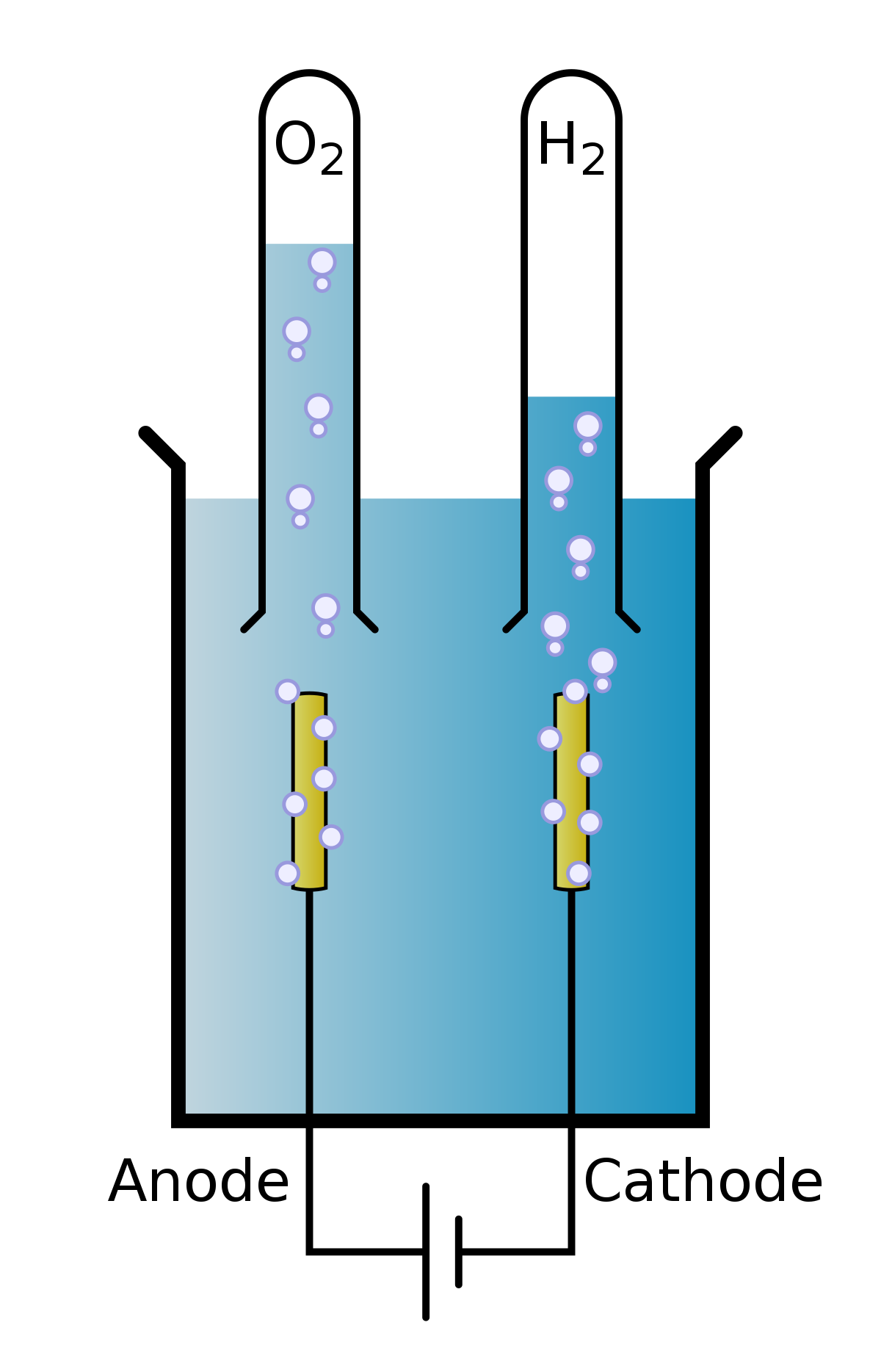 Source: simple.wikipedia.org
Source: simple.wikipedia.org
Applications since hydrogen can be produced more affordably from fossil fuels. Applications since hydrogen can be produced more affordably from fossil fuels. Hydrogen gas released in this way can be used as hydrogen fuel or remixed with the oxygen to create oxyhydrogen gas which is used in welding and other applications. Electrolysis of water produces hydrogen gas at the cathode which contains a lower proportion of deuterium than the original water. Electrolysis of water experiment energy is stored in the bonds of molecules.
 Source: docbrown.info
Source: docbrown.info
Electrolysis of water is the process of using electricity to decompose water into oxygen and hydrogen gas. Electrolysis of water. Compound can be extracted from water electrolysis will be able to explain how energy hydrogen flows through the electrolysis system molecule oxygen. Sometimes called water splitting electrolysis requires a minimum potential difference of 1 23 volts. When these bonds split apart the energy released can be used to do work.
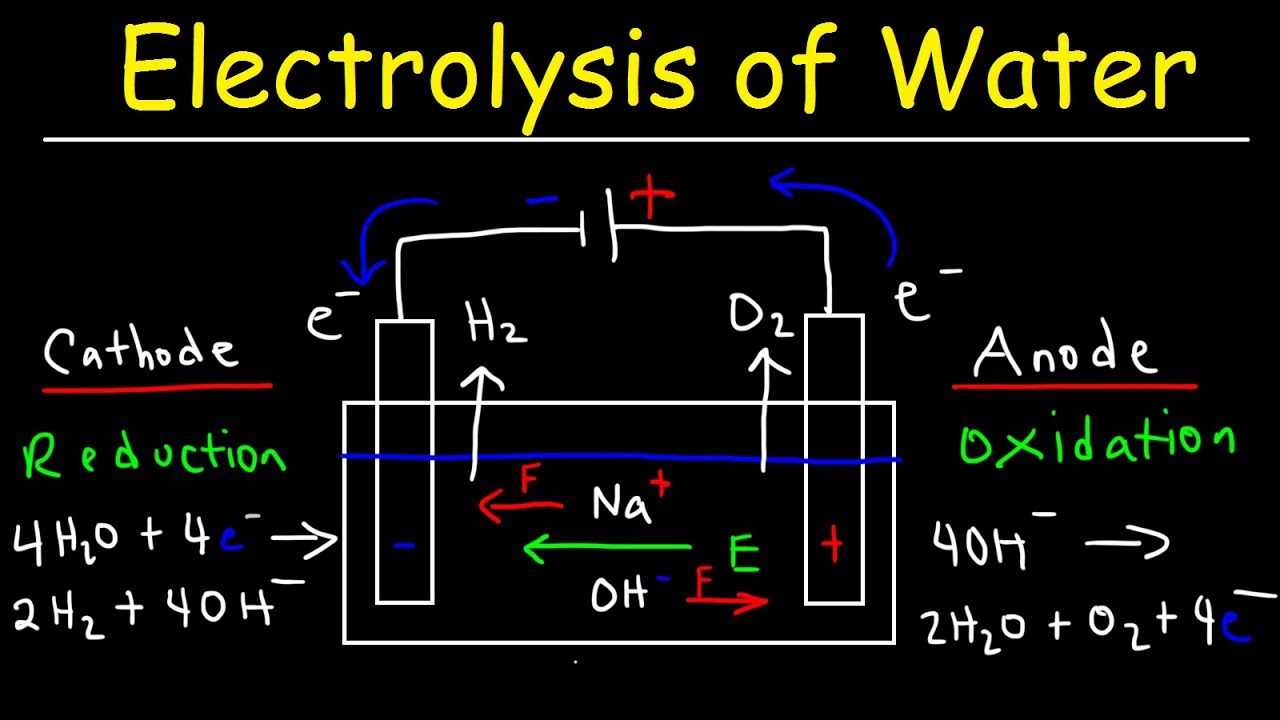 Source: youtube.com
Source: youtube.com
Breaking apart liquid water molecules into hydrogen and oxygen gas creates an enormous amount of energy which can be turned into useful electricity to power our homes and cars. Applications since hydrogen can be produced more affordably from fossil fuels. What is electrolysisthe electrolytic process of deposition of superior metal on the surface of the base metal or article is known as electro platingit makes. Electrolysis of water. A non spontaneous reaction is one that needs energy to work while it proceeds.
 Source: toppr.com
Source: toppr.com
Photovoltaic cell 3v min or 9 volt. Electrolysis of water experiment energy is stored in the bonds of molecules. Electrolysis of water produces hydrogen gas at the cathode which contains a lower proportion of deuterium than the original water. Will be able to explain how hydrogen. Electrolysis is the chemical process of using an electrical current to stimulate non spontaneous reactions.
 Source: docbrown.info
Source: docbrown.info
Battery 1 per group. A non spontaneous reaction is one that needs energy to work while it proceeds. Electrolysis of water. Battery 1 per group. Breaking apart liquid water molecules into hydrogen and oxygen gas creates an enormous amount of energy which can be turned into useful electricity to power our homes and cars.
If you find this site helpful, please support us by sharing this posts to your favorite social media accounts like Facebook, Instagram and so on or you can also save this blog page with the title electrolysis of water explained by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.





