Lemon and potato battery experiment
If you’re looking for lemon and potato battery experiment images information linked to the lemon and potato battery experiment keyword, you have come to the right blog. Our website frequently provides you with hints for seeking the maximum quality video and picture content, please kindly hunt and locate more enlightening video content and graphics that fit your interests.
Lemon And Potato Battery Experiment. If there is time replace the lemon with an apple or a potato and repeat. It can be done in various forms for different grades. So now we have made both a lemon battery and a potato battery which one is better. This experiment shows how a wet cell battery works.
 Build Potato Battery From bristolwatch.com
Build Potato Battery From bristolwatch.com
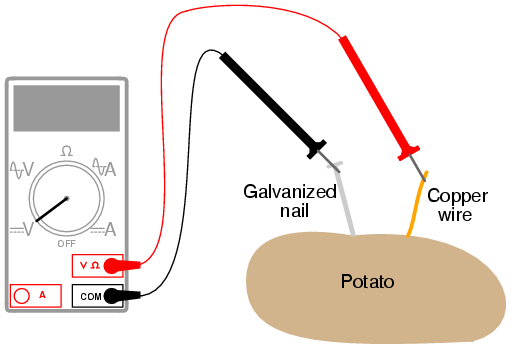
Building an electro chemical battery with a lemon is the classic science fair experiment. Research and explain how lemons and potatoes convert chemical to electrical energy. A lemon battery consists of two metals suspended in an acidic solution. Although you can use almost any fruit or potato all you need is one galvanized nail and a single copper penny although using a copper nail works better. Several potatoes galvanised nails or zink wire foil. Following materials and equipment required for the project.
In our experiment we needed electrodes to produce energy the anode was zinc the cathode was copper and the electrolyte was the lemon juice potato juice.
Chemicals in the fruit or vegetable create a negative charge in the zinc strip. The links will take you to that experiment if you would like to review how it relates to the lemon battery first. Copper and zinc work well as metals for the battery and the citric acid present in the lemon act as an acidic solution. However the potato battery was definitely a lot more work. The lemon battery experiment is one of the common experiments mentioned in science textbooks. Several potatoes galvanised nails or zink wire foil.
 Source: pinterest.com
Source: pinterest.com
Both were able to light up our led light bulbs so in that sense they are both successful. Attach one clip to the galvanized nail in the first potato and the other clip to the copper coin in the second potato. Copper and zinc work well as metals for the battery and the citric acid present in the lemon act as an acidic solution. A lemon battery consists of two metals suspended in an acidic solution. Research and explain how lemons and potatoes convert chemical to electrical energy.
 Source: bristolwatch.com
Source: bristolwatch.com
The lemon and the potato act like a low power battery. The lemon battery experiment is one of the common experiments mentioned in science textbooks. Although you can use almost any fruit or potato all you need is one galvanized nail and a single copper penny although using a copper nail works better. If you demonstrated the experiment try to give the students some time to do the lemon battery project themselves. Electrons move into the zinc strip and travel up the wire attached.
 Source: indiamart.com
Source: indiamart.com
This experiment shows how a wet cell battery works. Finishing this step the two potatoes should be attached to each other and the clock. Attach one clip to the galvanized nail in the first potato and the other clip to the copper coin in the second potato. Both were able to light up our led light bulbs so in that sense they are both successful. It can be done in various forms for different grades.
 Source: ameliascience7.weebly.com
Source: ameliascience7.weebly.com
The lemon and the potato act like a low power battery. In my experiment we wanted to see if we could convert energy using lemons and potatoes. Building an electro chemical battery with a lemon is the classic science fair experiment. In our experiment we needed electrodes to produce energy the anode was zinc the cathode was copper and the electrolyte was the lemon juice potato juice. So if you are looking for a quicker experiment the lemon battery is faster and easier.
 Source: m.youtube.com
Source: m.youtube.com
In our experiment we needed electrodes to produce energy the anode was zinc the cathode was copper and the electrolyte was the lemon juice potato juice. So if you are looking for a quicker experiment the lemon battery is faster and easier. Although you can use almost any fruit or potato all you need is one galvanized nail and a single copper penny although using a copper nail works better. In my experiment we wanted to see if we could convert energy using lemons and potatoes. If you demonstrated the experiment try to give the students some time to do the lemon battery project themselves.
 Source: sciencewithkids.com
Source: sciencewithkids.com
It can be done in various forms for different grades. In my experiment we wanted to see if we could convert energy using lemons and potatoes. Several potatoes galvanised nails or zink wire foil. A lemon battery consists of two metals suspended in an acidic solution. It can be done in various forms for different grades.
 Source: sciencewithkids.com
Source: sciencewithkids.com
Building an electro chemical battery with a lemon is the classic science fair experiment. Research and explain how lemons and potatoes convert chemical to electrical energy. It s fun and safe. So now we have made both a lemon battery and a potato battery which one is better. Copper and zinc work well as metals for the battery and the citric acid present in the lemon act as an acidic solution.
 Source: sciencewithkids.com
Source: sciencewithkids.com
Chemicals in the fruit or vegetable create a negative charge in the zinc strip. The lemon and the potato act like a low power battery. However the potato battery was definitely a lot more work. It can be done in various forms for different grades. In my experiment we wanted to see if we could convert energy using lemons and potatoes.
 Source: en.wikipedia.org
Source: en.wikipedia.org
A lemon battery consists of two metals suspended in an acidic solution. A lemon battery consists of two metals suspended in an acidic solution. If there is time replace the lemon with an apple or a potato and repeat. This experiment shows how a wet cell battery works. So if you are looking for a quicker experiment the lemon battery is faster and easier.
 Source: ameliascience7.weebly.com
Source: ameliascience7.weebly.com
According to naked scientists contributor dave ansell their lemon battery would have required 5 000 hours to charge their battery and he predicted it would have most likely died within a mere 30 minutes. So now we have made both a lemon battery and a potato battery which one is better. This will complete the circuit for your battery. It can be done in various forms for different grades. Although you can use almost any fruit or potato all you need is one galvanized nail and a single copper penny although using a copper nail works better.
 Source: varvarag.wordpress.com
Source: varvarag.wordpress.com
Both were able to light up our led light bulbs so in that sense they are both successful. If you demonstrated the experiment try to give the students some time to do the lemon battery project themselves. According to naked scientists contributor dave ansell their lemon battery would have required 5 000 hours to charge their battery and he predicted it would have most likely died within a mere 30 minutes. First we had to roll out our lemons and open the juice pockets the more juice their is the better. This experiment shows how a wet cell battery works.
 Source: ameliascience7.weebly.com
Source: ameliascience7.weebly.com
So now we have made both a lemon battery and a potato battery which one is better. So now we have made both a lemon battery and a potato battery which one is better. Several potatoes galvanised nails or zink wire foil. In our experiment we needed electrodes to produce energy the anode was zinc the cathode was copper and the electrolyte was the lemon juice potato juice. This will complete the circuit for your battery.
 Source: kit4curious.com
Source: kit4curious.com
A lemon battery consists of two metals suspended in an acidic solution. It s fun and safe. Attach one clip to the galvanized nail in the first potato and the other clip to the copper coin in the second potato. If there is time replace the lemon with an apple or a potato and repeat. This experiment shows how a wet cell battery works.
 Source: youtube.com
Source: youtube.com
Several potatoes galvanised nails or zink wire foil. If there is time replace the lemon with an apple or a potato and repeat. The links will take you to that experiment if you would like to review how it relates to the lemon battery first. So now we have made both a lemon battery and a potato battery which one is better. According to naked scientists contributor dave ansell their lemon battery would have required 5 000 hours to charge their battery and he predicted it would have most likely died within a mere 30 minutes.
 Source: sciencewithkids.com
Source: sciencewithkids.com
Although you can use almost any fruit or potato all you need is one galvanized nail and a single copper penny although using a copper nail works better. The lemon and the potato act like a low power battery. Connect the two potatoes with one alligator clip lead. In our experiment we needed electrodes to produce energy the anode was zinc the cathode was copper and the electrolyte was the lemon juice potato juice. Several potatoes galvanised nails or zink wire foil.
If you find this site helpful, please support us by sharing this posts to your preference social media accounts like Facebook, Instagram and so on or you can also save this blog page with the title lemon and potato battery experiment by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.