Salt water energy explanation
If you’re searching for salt water energy explanation pictures information linked to the salt water energy explanation interest, you have pay a visit to the ideal site. Our site always gives you suggestions for seeing the highest quality video and picture content, please kindly surf and locate more informative video articles and graphics that match your interests.
Salt Water Energy Explanation. The key waste product is brackish water. Production of energy from the difference between salt water and fresh water is most convenient near the oceans but now using an ammonium bicarbonate salt solution researchers can combine. Two practical methods for this are reverse electrodialysis and pressure retarded osmosis. M per second is available.
 Salt Water Energy By From prezi.com
Salt Water Energy By From prezi.com
The flow of fresh water into seas that are made up of salt water. The temperature needed to boil will increase about 0 5 c for every 58 grams of dissolved salt per kilogram of water. Salt water with a substantial amount of salt dissolved in it is chemically and physically quite distinct from fresh water. Place a zinc coated nail into the solution and tape it to one side of the cup securely. Two practical methods for this are reverse electrodialysis and pressure retarded osmosis. Make a saltwater solution by mixing a small jar of water with a teaspoon of salt.
Production of energy from the difference between salt water and fresh water is most convenient near the oceans but now using an ammonium bicarbonate salt solution researchers can combine.
At 2 4v the battery s specific energy was approximately 100 watt hour kg and it displayed consistent performance over 1 000 charge discharge cycles. It raised the maximum voltage for such a battery from 1 23 v to around 3 v. First salt water has substantially higher density enough that in very salty water like the dead sea or the great salt lake it is very difficult to dive human bodies float substantially higher in the water. The flow of fresh water into seas that are made up of salt water. The key waste product is brackish water. It occurs any time you add a nonvolatile solute such as salt to a solvent such as water.
 Source: prezi.com
Source: prezi.com
Production of energy from the difference between salt water and fresh water is most convenient near the oceans but now using an ammonium bicarbonate salt solution researchers can combine. This byproduct is the result of natural forces that are being harnessed. This is an example of boiling point elevation and it is not exclusive to water. Place a zinc coated nail into the solution and tape it to one side of the cup securely. Guys are you want to make energy fine what you need is salt.
 Source: electronicproducts.com
Source: electronicproducts.com
These are present in sea water and its possible releasing them to the environment could be hazardous to humans and to wildlife and plant matter. What about sodium sulfates etc. To be useful in the real world salt water energy must be an energy positive process. Osmotic power salinity gradient power or blue energy is the energy available from the difference in the salt concentration between seawater and river water. No one has claimed this is the case.
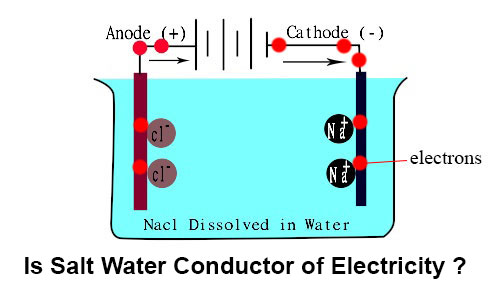
 Source: masterconceptsinchemistry.com
Source: masterconceptsinchemistry.com
It takes a lot of energy to break apart an ionic bond and the stronger the attraction the stronger the bond. Explanation examples. No one has claimed this is the case. Ohh not the movieeethe white powder you use in your kitchenyes the salt my last year science project now what you need are1. Both processes rely on osmosis with membranes.
 Source: teachengineering.org
Source: teachengineering.org
Production of energy from the difference between salt water and fresh water is most convenient near the oceans but now using an ammonium bicarbonate salt solution researchers can combine. It raised the maximum voltage for such a battery from 1 23 v to around 3 v. Guys are you want to make energy fine what you need is salt. At 2 4v the battery s specific energy was approximately 100 watt hour kg and it displayed consistent performance over 1 000 charge discharge cycles. Both processes rely on osmosis with membranes.
 Source: slideshare.net
Source: slideshare.net
Make a saltwater solution by mixing a small jar of water with a teaspoon of salt. This byproduct is the result of natural forces that are being harnessed. Acidic basic salt solutions. Production of energy from the difference between salt water and fresh water is most convenient near the oceans but now using an ammonium bicarbonate salt solution researchers can combine. Explanation examples.

The high salt concentration allows the interphase to form. This will be the negative electrode. Ohh not the movieeethe white powder you use in your kitchenyes the salt my last year science project now what you need are1. As well as the ability. Salt water energy uses the hydrogen in seawater but what happens if chloride is released in the process.
 Source: youtube.com
Source: youtube.com
If you add salt to water you raise the water s boiling point or the temperature at which it will boil. It occurs any time you add a nonvolatile solute such as salt to a solvent such as water. The key waste product is brackish water. To be useful in the real world salt water energy must be an energy positive process. Place a copper coated wire into the solution and tape it to the other side of the cup securely.
 Source: en.wikipedia.org
Source: en.wikipedia.org
Acidic basic salt solutions. It occurs any time you add a nonvolatile solute such as salt to a solvent such as water. Osmotic power salinity gradient power or blue energy is the energy available from the difference in the salt concentration between seawater and river water. This is an example of boiling point elevation and it is not exclusive to water. Place a copper coated wire into the solution and tape it to the other side of the cup securely.
 Source: scholastic.com
Source: scholastic.com
Two practical methods for this are reverse electrodialysis and pressure retarded osmosis. These are present in sea water and its possible releasing them to the environment could be hazardous to humans and to wildlife and plant matter. This byproduct is the result of natural forces that are being harnessed. This will be the negative electrode. At 2 4v the battery s specific energy was approximately 100 watt hour kg and it displayed consistent performance over 1 000 charge discharge cycles.
 Source: slideshare.net
Source: slideshare.net
Explanation examples. This is an example of boiling point elevation and it is not exclusive to water. In 1954 pattle suggested. Salt water with a substantial amount of salt dissolved in it is chemically and physically quite distinct from fresh water. To be useful in the real world salt water energy must be an energy positive process.
 Source: plethorist.com
Source: plethorist.com
Ohh not the movieeethe white powder you use in your kitchenyes the salt my last year science project now what you need are1. M per second is available. Place a copper coated wire into the solution and tape it to the other side of the cup securely. This is an example of boiling point elevation and it is not exclusive to water. Production of energy from the difference between salt water and fresh water is most convenient near the oceans but now using an ammonium bicarbonate salt solution researchers can combine.
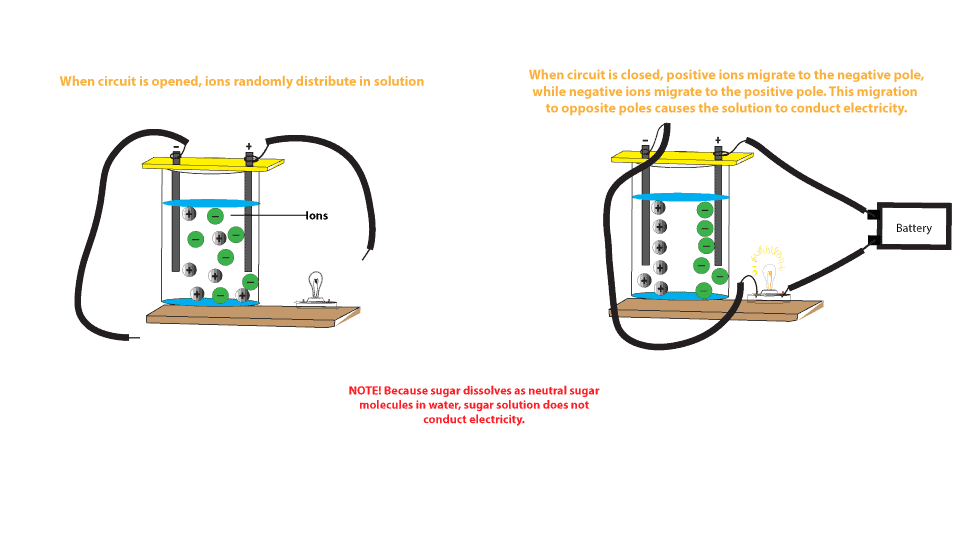
 Source: slideshare.net
Source: slideshare.net
Make a saltwater solution by mixing a small jar of water with a teaspoon of salt. What about sodium sulfates etc. At 2 4v the battery s specific energy was approximately 100 watt hour kg and it displayed consistent performance over 1 000 charge discharge cycles. The high salt concentration allows the interphase to form. Salt water energy uses the hydrogen in seawater but what happens if chloride is released in the process.
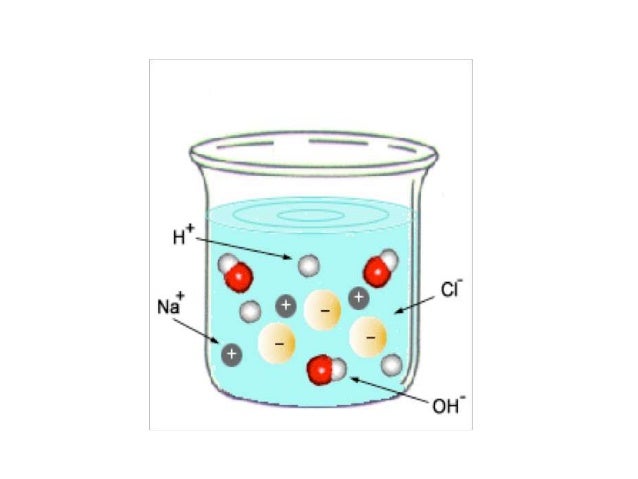
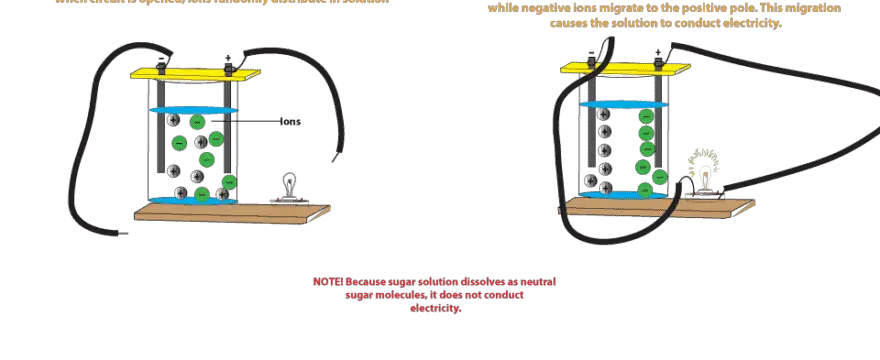 Source: masterconceptsinchemistry.com
Source: masterconceptsinchemistry.com
It occurs any time you add a nonvolatile solute such as salt to a solvent such as water. The flow of fresh water into seas that are made up of salt water. These are present in sea water and its possible releasing them to the environment could be hazardous to humans and to wildlife and plant matter. This is an example of boiling point elevation and it is not exclusive to water. Two practical methods for this are reverse electrodialysis and pressure retarded osmosis.
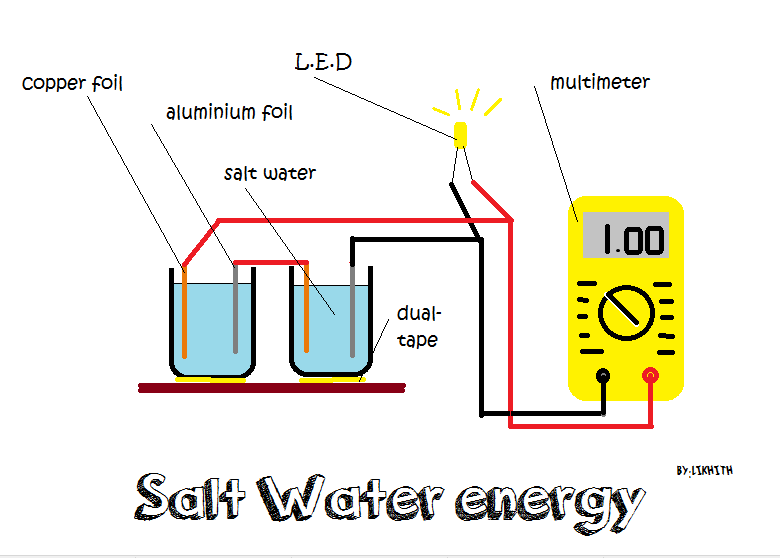 Source: instructables.com
Source: instructables.com
To be useful in the real world salt water energy must be an energy positive process. Salt water energy uses the hydrogen in seawater but what happens if chloride is released in the process. As well as the ability. M per second is available. The temperature needed to boil will increase about 0 5 c for every 58 grams of dissolved salt per kilogram of water.
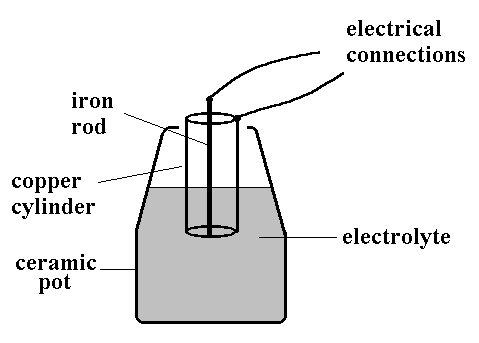 Source: creative-science.org.uk
Source: creative-science.org.uk
Place a copper coated wire into the solution and tape it to the other side of the cup securely. Osmotic power salinity gradient power or blue energy is the energy available from the difference in the salt concentration between seawater and river water. Acidic basic salt solutions. Both processes rely on osmosis with membranes. This byproduct is the result of natural forces that are being harnessed.
If you find this site convienient, please support us by sharing this posts to your preference social media accounts like Facebook, Instagram and so on or you can also bookmark this blog page with the title salt water energy explanation by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.