Splitting water electrolysis experiment
If you’re looking for splitting water electrolysis experiment images information linked to the splitting water electrolysis experiment keyword, you have visit the ideal site. Our website always provides you with hints for downloading the maximum quality video and image content, please kindly hunt and locate more informative video content and images that fit your interests.
Splitting Water Electrolysis Experiment. Experiment 3 demonstrates the concept of electrolysis. The chemical change occurs when the substance loses electrons oxidation or gains them reduction. Electrolysis experiments video. Water electrolysis splitting experiment hypothesis.
 Splitting Water Using Electrolysis News About Energy Storage Batteries Climate Change And The Environment From upsbatterycenter.com
Splitting Water Using Electrolysis News About Energy Storage Batteries Climate Change And The Environment From upsbatterycenter.com
This experiment has significant implications in terms of what these 2 gases can be used for in their own right with hydrogen being one of the cleanest sources of energy we have access to. Splitting water electrolysis chemical reactions occur when two or more elements or compounds interact with each other to form new products. Electrolysis experiments video. As for nitrous nox it is not a fuel it is an oxidizer. When these bonds split apart the energy released can be used to do work. Electrolysis of water experiment energy is stored in the bonds of molecules.
The process of splitting water h 2 o into its atomic components hydrogen and oxygen using electricity is known as electrolysis.
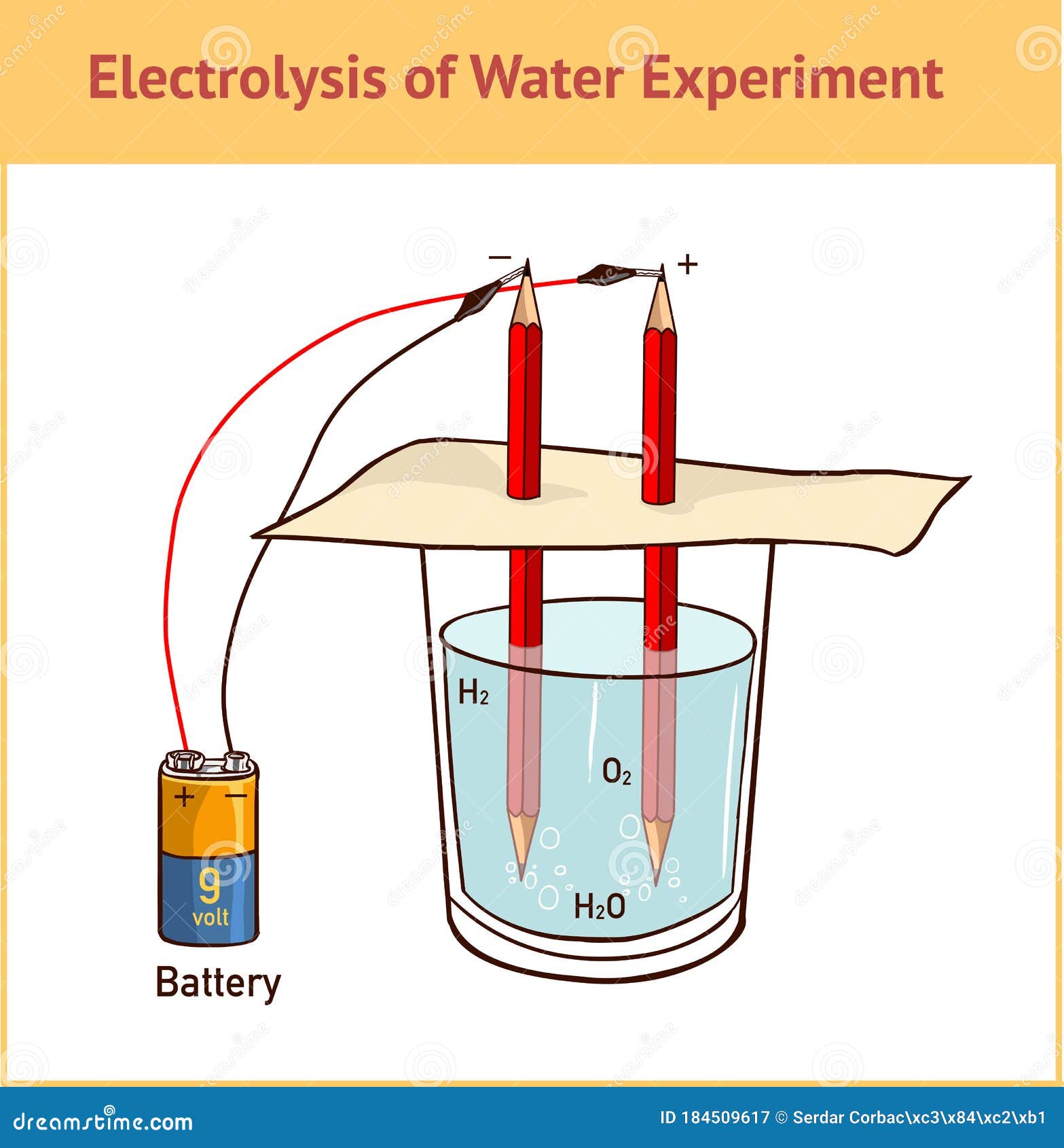
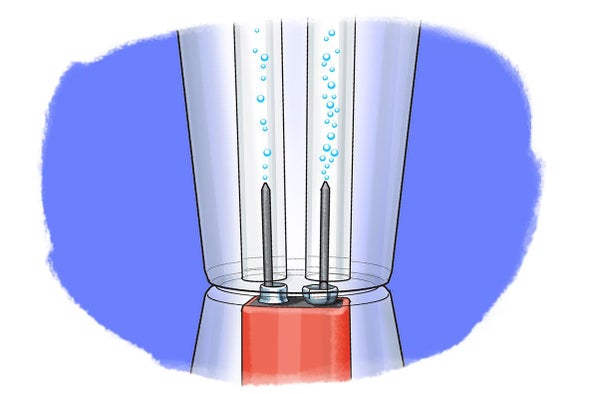
On passing electricity through water it splits or electrolyzes to give off hydrogen and oxygen gases at the two electrodes. Dissolve the bicarb of soda in the water and fill the tub. Any gases collected during the electrolysis will replace the water in the tubes so make sure there are no air bubbles. 6 or 9 volt battery. Hi guys watch this amazing video electrolysis of water experiment h2o electrolysis electrolysis water electrolysis chemistry like it and comments below. Stanley meyer s secret method went with him to his grave.
 Source: youtube.com
Source: youtube.com
Breaking apart liquid water molecules into hydrogen and oxygen gas creates an enormous amount of energy which can be turned into useful electricity to power our homes and cars. It all started with stanley meyer which claimed he was able to run a car just on water. Breaking apart liquid water molecules into hydrogen and oxygen gas creates an enormous amount of energy which can be turned into useful electricity to power our homes and cars. Finally remove each test tube fill it with the water and carefully replace it over the graphite. When these bonds split apart the energy released can be used to do work.
 Source: dreamstime.com
Source: dreamstime.com
As for nitrous nox it is not a fuel it is an oxidizer. As for nitrous nox it is not a fuel it is an oxidizer. Electrolysis is the process by which an electric current is passed through a substance to affect a chemical change. Dissolve the bicarb of soda in the water and fill the tub. This includes for example how we digest food or produce energy.
 Source: scienceprojectideas.org
Source: scienceprojectideas.org
And let them dream of ways to use and improve the knowledge. Let them do their experiments and let them form their hypothesis. In nature reactions often occur as part of a more complex set of reactions. Electrolysis enthusiasts believe that pulsed direct current at specific frequencies increases the efficiency of splitting water into it s components. When these bonds split apart the energy released can be used to do work.
 Source: upsbatterycenter.com
Source: upsbatterycenter.com
Electrolysis experiments video. The process of splitting water h 2 o into its atomic components hydrogen and oxygen using electricity is known as electrolysis. Let them do their experiments and let them form their hypothesis. Experiment 3 demonstrates the concept of electrolysis. As for nitrous nox it is not a fuel it is an oxidizer.
 Source: nigerianscholars.com
Source: nigerianscholars.com
Piece of thin cardstock or cardboard. Electrolysis enthusiasts believe that pulsed direct current at specific frequencies increases the efficiency of splitting water into it s components. In the two experiments listed below the first reactive substance is water and the. On passing electricity through water it splits or electrolyzes to give off hydrogen and oxygen gases at the two electrodes. Electrolysis is the process by which an electric current is passed through a substance to affect a chemical change.
 Source: esmartkids.epcor.ca
Source: esmartkids.epcor.ca
In the two experiments listed below the first reactive substance is water and the. 6 or 9 volt battery. Breaking apart liquid water molecules into hydrogen and oxygen gas creates an enormous amount of energy which can be turned into useful electricity to power our homes and cars. When these bonds split apart the energy released can be used to do work. On passing electricity through water it splits or electrolyzes to give off hydrogen and oxygen gases at the two electrodes.
 Source: scientificamerican.com
Source: scientificamerican.com
Let them do their experiments and let them form their hypothesis. Energy is stored in the bonds of molecules. As for nitrous nox it is not a fuel it is an oxidizer. This experiment has significant implications in terms of what these 2 gases can be used for in their own right with hydrogen being one of the cleanest sources of energy we have access to. In the two experiments listed below the first reactive substance is water and the.
 Source: studylib.net
Source: studylib.net
Let them do their experiments and let them form their hypothesis. The chemical change occurs when the substance loses electrons oxidation or gains them reduction. Electrolysis of water experiment energy is stored in the bonds of molecules. Stanley meyer s secret method went with him to his grave. Breaking apart liquid water molecules into hydrogen and oxygen gas creates an enormous amount of energy which can be turned into useful electricity to power our homes and cars.
 Source: learning-center.homesciencetools.com
Source: learning-center.homesciencetools.com
Finally remove each test tube fill it with the water and carefully replace it over the graphite. Finally remove each test tube fill it with the water and carefully replace it over the graphite. The process of splitting water h 2 o into its atomic components hydrogen and oxygen using electricity is known as electrolysis. When these bonds split apart the energy released can be used to do work. Dissolve the bicarb of soda in the water and fill the tub.
 Source: en.wikipedia.org
Source: en.wikipedia.org
Electrolysis enthusiasts believe that pulsed direct current at specific frequencies increases the efficiency of splitting water into it s components. And let them dream of ways to use and improve the knowledge. Oxides are what make the existing fuel burn hotter. Stanley meyer s secret method went with him to his grave. 6 or 9 volt battery.
 Source: pinterest.com
Source: pinterest.com
This includes for example how we digest food or produce energy. Breaking apart liquid water molecules into hydrogen and oxygen gas creates an enormous amount of energy which can be turned into useful electricity to power our homes and cars. The process of splitting water h 2 o into its atomic components hydrogen and oxygen using electricity is known as electrolysis. In nature reactions often occur as part of a more complex set of reactions. Electrolysis of water is the easiest way to demonstrate the chemical process of separating the ions.
 Source: orbitingfrog.com
Source: orbitingfrog.com
In the two experiments listed below the first reactive substance is water and the. Two insulated wires or alligator clip leads. Electrolysis of water experiment energy is stored in the bonds of molecules. Any gases collected during the electrolysis will replace the water in the tubes so make sure there are no air bubbles. As for nitrous nox it is not a fuel it is an oxidizer.
 Source: upsbatterycenter.com
Source: upsbatterycenter.com
Breaking apart liquid water molecules into hydrogen and oxygen gas creates an enormous amount of energy which can be turned into useful electricity to power our homes and cars. Breaking apart liquid water molecules into hydrogen and oxygen gas creates an enormous amount of energy which can be turned into useful electricity to power our homes and cars. Let them do their experiments and let them form their hypothesis. Water electrolysis splitting experiment hypothesis. This experiment has significant implications in terms of what these 2 gases can be used for in their own right with hydrogen being one of the cleanest sources of energy we have access to.
 Source: orbitingfrog.com
Source: orbitingfrog.com
Oxides are what make the existing fuel burn hotter. In nature reactions often occur as part of a more complex set of reactions. Finally remove each test tube fill it with the water and carefully replace it over the graphite. Splitting water electrolysis chemical reactions occur when two or more elements or compounds interact with each other to form new products. Hi guys watch this amazing video electrolysis of water experiment h2o electrolysis electrolysis water electrolysis chemistry like it and comments below.
 Source: navigatingbyjoy.com
Source: navigatingbyjoy.com
6 or 9 volt battery. Stanley meyer s secret method went with him to his grave. Finally remove each test tube fill it with the water and carefully replace it over the graphite. Piece of thin cardstock or cardboard. In the two experiments listed below the first reactive substance is water and the.
If you find this site helpful, please support us by sharing this posts to your own social media accounts like Facebook, Instagram and so on or you can also save this blog page with the title splitting water electrolysis experiment by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.