Sulphur and iron filings experiment
If you’re looking for sulphur and iron filings experiment images information linked to the sulphur and iron filings experiment keyword, you have visit the ideal site. Our website frequently gives you hints for viewing the maximum quality video and picture content, please kindly surf and locate more enlightening video content and images that fit your interests.
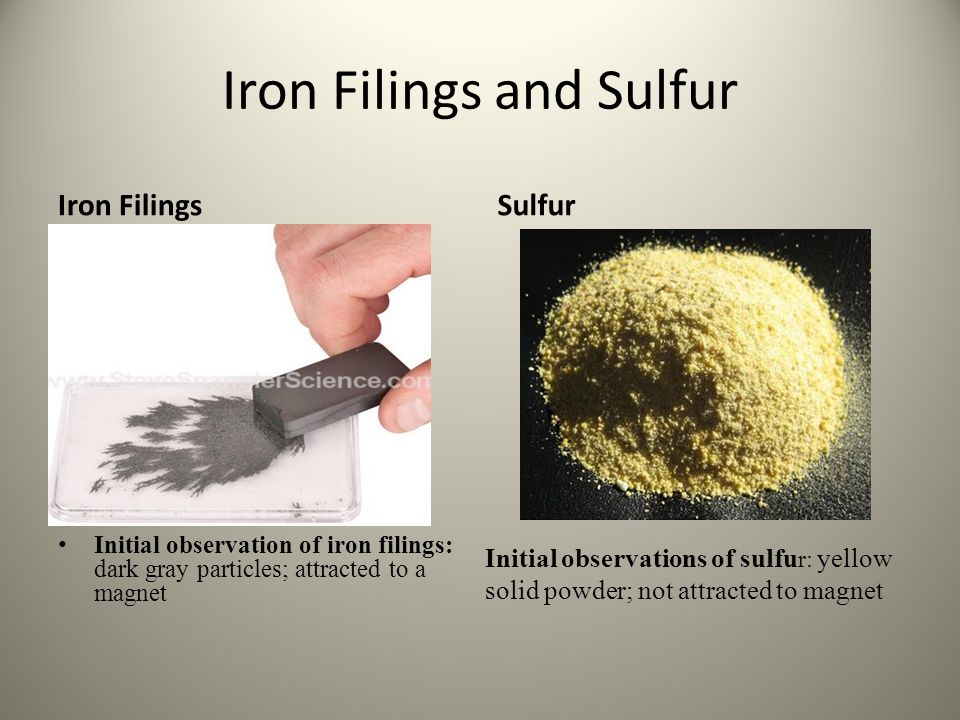
Sulphur And Iron Filings Experiment. Mix the two powders by pouring repeatedly from one piece of paper to the other until a homogeneous mixture by appearance is obtained. Neodymium is a rare earth metal. Iron filings reacts with sulphur to make iron sulphide fes not iron sulphate feso4. There is the smell of rotten eggs in the air that the sulfur is releasing as it is heated.
 To Prepare A Mixture A Compound And Distinguish Them On The Basis Of Appearance Behaviour Towards Youtube From youtube.com
To Prepare A Mixture A Compound And Distinguish Them On The Basis Of Appearance Behaviour Towards Youtube From youtube.com
The quantity seemed less to the naked eyes but after he saw the weight he was surprised. The iron is magnetic and is attracted to the magnet while sulphur being non magnetic is not attracted to the magnet. He put them on a china dish and heated it till the mixture became red hot. Do this by weighing out 7 g of iron powder and 4 g of finely powdered sulfur onto separate pieces of filter paper or use weighing boats. A mixture of iron fillings and sulphur. Do an experiment on iron filings and sulphur.
Iron filings reacts with sulphur to make iron sulphide fes not iron sulphate feso4.
Part a was heated strongly while part b was not heated. Close up footage of a mixture of iron filings grey and sulphur powder yellow being separated using a bar magnet. The reaction is fe s fes. The quantity seemed less to the naked eyes but after he saw the weight he was surprised. Iron filings and sulphur were mixed together and divided into two parts a and b. This is a demonstration of magnetism.
 Source: slideplayer.com
Source: slideplayer.com
Iron filings reacts with sulphur to make iron sulphide fes not iron sulphate feso4. Mix the two powders by pouring repeatedly from one piece of paper to the other until a homogeneous mixture by appearance is obtained. Iron fillings powdered sulphur dilute hydrochloric acid piece of wood piece of paper bar magnet 2 test tubes 2 beakers stopper water. Do an experiment on iron filings and sulphur. 56 g of iron will fully react with 32 g of sulphur to produce 88 2 x g of.

A mixture of iron fillings and sulphur. The iron is magnetic and is attracted to the magnet while sulphur being non magnetic is not attracted to the magnet. This is a demonstration of magnetism. C dilute hydrochloric acid was added to both the parts and evolution of gas was seen in. He put them on a china dish and heated it till the mixture became red hot.
 Source: sciencephoto.com
Source: sciencephoto.com
First he took 3 g of sulphur powder and 5 g of iron filings. Iron filings and sulphur were mixed together and divided into two parts a and b. First he took 3 g of sulphur powder and 5 g of iron filings. Then he let the mixture cool and weighed the mixture. Close up footage of a mixture of iron filings grey and sulphur powder yellow being separated using a bar magnet.
 Source: sites.google.com
Source: sites.google.com
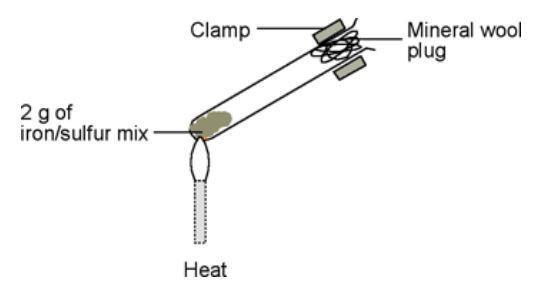
Sulfur dioxide so 2 g toxic is formed if the sulfur catches fire see cleapss hazcard hc097. B carbon disulphide was added to both a and b. Then he let the mixture cool and weighed the mixture. Iron filings heated with sulfur powder chemical reaction in a chemical reaction between iron filings and sulfur powder the physical properties of iron whic. Let s look at the first experiment involving the mixture of iron fillings and sulphur.
 Source: alamy.com
Source: alamy.com
He put them on a china dish and heated it till the mixture became red hot. Iron filings heated with sulfur powder chemical reaction in a chemical reaction between iron filings and sulfur powder the physical properties of iron whic. The quantity seemed less to the naked eyes but after he saw the weight he was surprised. This is a demonstration of magnetism. 56 g of iron will fully react with 32 g of sulphur to produce 88 2 x g of.
 Source: sites.google.com
Source: sites.google.com
Iron filings and sulphur were mixed together and divided into two parts a and b. B carbon disulphide was added to both a and b. Iron filings reacts with sulphur to make iron sulphide fes not iron sulphate feso4. Let s look at the first experiment involving the mixture of iron fillings and sulphur. The strongest in the world supermagnete.
 Source: 20304joycechancs.blogspot.com
Source: 20304joycechancs.blogspot.com
Iron filings reacts with sulphur to make iron sulphide fes not iron sulphate feso4. A magnet was passed over both a and b. A mixture of iron fillings and sulphur. C dilute hydrochloric acid was added to both the parts and evolution of gas was seen in. Then he let the mixture cool and weighed the mixture.
 Source: slideplayer.com
Source: slideplayer.com
Let s look at the first experiment involving the mixture of iron fillings and sulphur. Write the observations when. It s poisonous sulfur dioxide which sulfur will become when chemically combined with oxygen so 2 the. Part a was heated strongly while part b was not heated. Mix the two powders by pouring repeatedly from one piece of paper to the other until a homogeneous mixture by appearance is obtained.
 Source: cbsesamplepapers.info
Source: cbsesamplepapers.info
A magnet was passed over both a and b. Part a was heated strongly while part b was not heated. A mixture of iron filings and sulphur. Iron filings heated with sulfur powder chemical reaction in a chemical reaction between iron filings and sulfur powder the physical properties of iron whic. Mix the two powders by pouring repeatedly from one piece of paper to the other until a homogeneous mixture by appearance is obtained.
 Source: slideplayer.com
Source: slideplayer.com
56 g of iron will fully react with 32 g of sulphur to produce 88 2 x g of. Iron filings and sulphur were mixed together and divided into two parts a and b. Part a was heated strongly while part b was not heated. The quantity seemed less to the naked eyes but after he saw the weight he was surprised. Iron filings heated with sulfur powder chemical reaction in a chemical reaction between iron filings and sulfur powder the physical properties of iron whic.
 Source: edu.rsc.org
Source: edu.rsc.org
First he took 3 g of sulphur powder and 5 g of iron filings. Mix the two powders by pouring repeatedly from one piece of paper to the other until a homogeneous mixture by appearance is obtained. The quantity seemed less to the naked eyes but after he saw the weight he was surprised. This is a demonstration of magnetism. Let s look at the first experiment involving the mixture of iron fillings and sulphur.
 Source: youtube.com
Source: youtube.com
Close up footage of a mixture of iron filings grey and sulphur powder yellow being separated using a bar magnet. It s poisonous sulfur dioxide which sulfur will become when chemically combined with oxygen so 2 the. Iron filings heated with sulfur powder chemical reaction in a chemical reaction between iron filings and sulfur powder the physical properties of iron whic. Neodymium is a rare earth metal. The strongest in the world supermagnete.
 Source: chemlegin.wordpress.com
Source: chemlegin.wordpress.com
Then he let the mixture cool and weighed the mixture. Then he let the mixture cool and weighed the mixture. Iron and sulphur magnetism experiment. Sulfur dioxide so 2 g toxic is formed if the sulfur catches fire see cleapss hazcard hc097. Iron filings reacts with sulphur to make iron sulphide fes not iron sulphate feso4.
 Source: youtube.com
Source: youtube.com
A magnet was passed over both a and b. First he took 3 g of sulphur powder and 5 g of iron filings. It s poisonous sulfur dioxide which sulfur will become when chemically combined with oxygen so 2 the. There is the smell of rotten eggs in the air that the sulfur is releasing as it is heated. He put them on a china dish and heated it till the mixture became red hot.
 Source: alamy.com
Source: alamy.com
Write the observations when. He put them on a china dish and heated it till the mixture became red hot. Iron filings reacts with sulphur to make iron sulphide fes not iron sulphate feso4. Let s look at the first experiment involving the mixture of iron fillings and sulphur. For this interesting and cool experiment you will need 00 00 neodymium magnets.
If you find this site value, please support us by sharing this posts to your favorite social media accounts like Facebook, Instagram and so on or you can also save this blog page with the title sulphur and iron filings experiment by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.





