Which best explains the surface tension of water
If you’re looking for which best explains the surface tension of water pictures information linked to the which best explains the surface tension of water interest, you have pay a visit to the right blog. Our website frequently provides you with suggestions for viewing the maximum quality video and picture content, please kindly surf and locate more informative video content and graphics that match your interests.
Which Best Explains The Surface Tension Of Water. If the surface is between two liquids such as water and oil it is called interface tension. Identify the true statements about surface tension. Why bubbles are round. Surface tension is the tendency of liquid surfaces to shrink into the minimum surface area possible.
 Values Of The Surface Tension Of Water At Different Temperatures 1 Download Table From researchgate.net
Values Of The Surface Tension Of Water At Different Temperatures 1 Download Table From researchgate.net
Which of these best explains the ability of small choose the substance with the lowest surface tension. If the surface is between two liquids such as water and oil it is called interface tension. Identify the true statements about surface tension. Why bubbles are round. Surface tension is the tendency of liquid surfaces to shrink into the minimum surface area possible. Surface tension is a phenomenon in which the surface of a liquid where the liquid is in contact with a gas acts as a thin elastic sheet.
Which of these best explains the ability of small choose the substance with the lowest surface tension.
Hope this helps. How do living things like insects use surface tension. Surface tension is the tendency of liquid surfaces to shrink into the minimum surface area possible. Collin2622 collin2622 it tends to aggregate in drops rather than spread out on a surface as a thin film. Which best explains the high surface tension of water get the answers you need now. Surface tension is responsible for the shape of liquid droplets.
 Source: thoughtco.com
Source: thoughtco.com
Why bubbles are round. Surface tension is responsible for the shape of liquid droplets. At liquid air interfaces surface tension results from the greater attraction of liquid molecules to each other due to cohesion than to the molecules in the. Which best explains the high surface tension of water get the answers you need now. Collin2622 collin2622 it tends to aggregate in drops rather than spread out on a surface as a thin film.
 Source: usgs.gov
Source: usgs.gov
Which statement best explains why tension grew why does water roll off the surface of. Surface tension and droplets. A high surface tension results from. Hope this helps. Surface tension is a phenomenon in which the surface of a liquid where the liquid is in contact with a gas acts as a thin elastic sheet.
 Source: doubtnut.com
Source: doubtnut.com
Surface tension allows insects e g. Surface tension is the tendency of liquid surfaces to shrink into the minimum surface area possible. Why bubbles are round. The tendency to minimize that wall tension pulls the bubbles into spherical shapes. How do living things like insects use surface tension.
 Source: researchgate.net
Source: researchgate.net
Surface tension and droplets. How do living things like insects use surface tension. Surface tension is responsible for the shape of liquid droplets. Surface tension is a phenomenon in which the surface of a liquid where the liquid is in contact with a gas acts as a thin elastic sheet. If the surface is between two liquids such as water and oil it is called interface tension.
 Source: doubtnut.com
Source: doubtnut.com
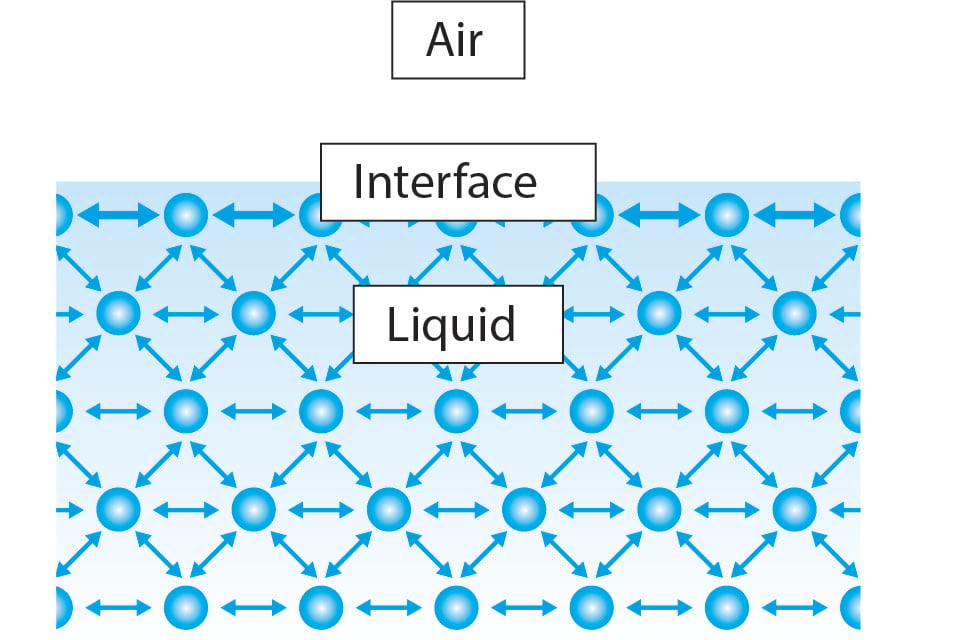
At liquid air interfaces surface tension results from the greater attraction of liquid molecules to each other due to cohesion than to the molecules in the. Surface tension is responsible for the shape of liquid droplets. Surface tension is the tendency of liquid surfaces to shrink into the minimum surface area possible. This term is typically used only when the liquid surface is in contact with gas such as the air. The tendency to minimize that wall tension pulls the bubbles into spherical shapes.
 Source: m.youtube.com
Source: m.youtube.com
The surface tension of water provides the necessary wall tension for the formation of bubbles with water. If the surface is between two liquids such as water and oil it is called interface tension. Surface tension is the tendency of liquid surfaces to shrink into the minimum surface area possible. This term is typically used only when the liquid surface is in contact with gas such as the air. Water striders to float and slide on a water surface without becoming even partly submerged.
 Source: biolinscientific.com
Source: biolinscientific.com
Surface tension allows insects e g. A high surface tension results from. At liquid air interfaces surface tension results from the greater attraction of liquid molecules to each other due to cohesion than to the molecules in the. Although easily deformed droplets of water tend to be pulled into a spherical shape by the cohesive forces of the surface layer. Which best explains the high surface tension of water get the answers you need now.
 Source: usgs.gov
Source: usgs.gov
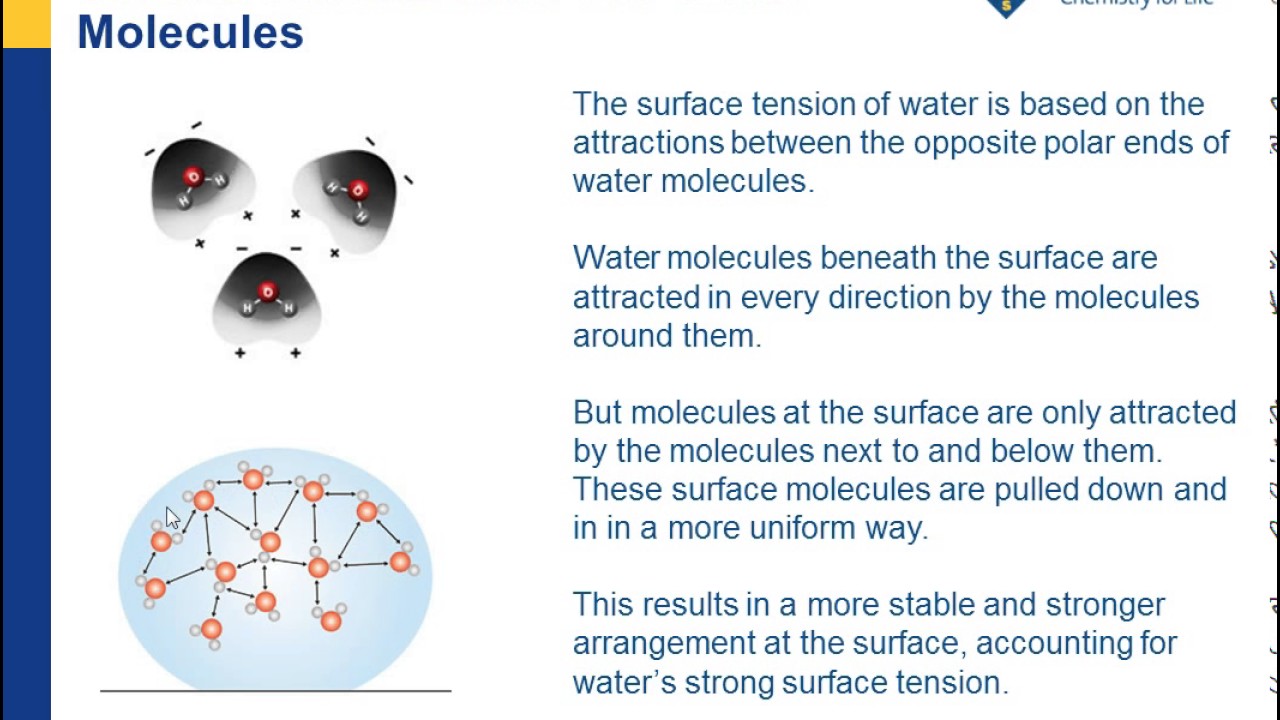
Which best explains the high surface tension of water 2 see answers photon photon its hydrogen bonding interactions. Collin2622 collin2622 it tends to aggregate in drops rather than spread out on a surface as a thin film. Which best explains the high surface tension of water get the answers you need now. Surface tension is the tendency of liquid surfaces to shrink into the minimum surface area possible. Surface tension is responsible for the shape of liquid droplets.
 Source: slideplayer.com
Source: slideplayer.com
Surface tension is the tendency of liquid surfaces to shrink into the minimum surface area possible. A high surface tension results from. Surface tension allows insects e g. Which best explains the high surface tension of water 2 see answers photon photon its hydrogen bonding interactions. The surface tension of water provides the necessary wall tension for the formation of bubbles with water.
 Source: kibron.com
Source: kibron.com
If the surface is between two liquids such as water and oil it is called interface tension. The surface tension of water provides the necessary wall tension for the formation of bubbles with water. Surface tension and droplets. A high surface tension results from. The tendency to minimize that wall tension pulls the bubbles into spherical shapes.
 Source: usgs.gov
Source: usgs.gov
Although easily deformed droplets of water tend to be pulled into a spherical shape by the cohesive forces of the surface layer. Which best explains the high surface tension of water get the answers you need now. Surface tension is a phenomenon in which the surface of a liquid where the liquid is in contact with a gas acts as a thin elastic sheet. The surface tension of water provides the necessary wall tension for the formation of bubbles with water. The tendency to minimize that wall tension pulls the bubbles into spherical shapes.
 Source: middleschoolchemistry.com
Source: middleschoolchemistry.com
Hope this helps. Surface tension and droplets. Which best explains the high surface tension of water 2 see answers photon photon its hydrogen bonding interactions. Why bubbles are round. This term is typically used only when the liquid surface is in contact with gas such as the air.
 Source: oneclass.com
Source: oneclass.com
Cohesion best explains the surface tension of water. Hope this helps. Why bubbles are round. Identify the true statements about surface tension. Which of these best explains the ability of small choose the substance with the lowest surface tension.
 Source: doubtnut.com
Source: doubtnut.com
The tendency to minimize that wall tension pulls the bubbles into spherical shapes. Which statement best explains why tension grew why does water roll off the surface of. How do living things like insects use surface tension. A high surface tension results from. Surface tension is responsible for the shape of liquid droplets.
 Source: usgs.gov
Source: usgs.gov
How do living things like insects use surface tension. Although easily deformed droplets of water tend to be pulled into a spherical shape by the cohesive forces of the surface layer. How do living things like insects use surface tension. This term is typically used only when the liquid surface is in contact with gas such as the air. Collin2622 collin2622 it tends to aggregate in drops rather than spread out on a surface as a thin film.
If you find this site convienient, please support us by sharing this posts to your preference social media accounts like Facebook, Instagram and so on or you can also bookmark this blog page with the title which best explains the surface tension of water by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.





